This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Step 1: Analyze the equilibrium reaction for Question 39. The given equilibrium reaction is: This indicates that the forward reaction (decomposition of PCl) is endothermic (absorbs heat). Also, the number of moles of gas increases from the reactant side (1 mole of PCl) to the product side (1 mole of PCl + 1 mole of Cl, totaling 2 moles of gas).
Step 2: Evaluate each statement for Question 39 based on Le Chat
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question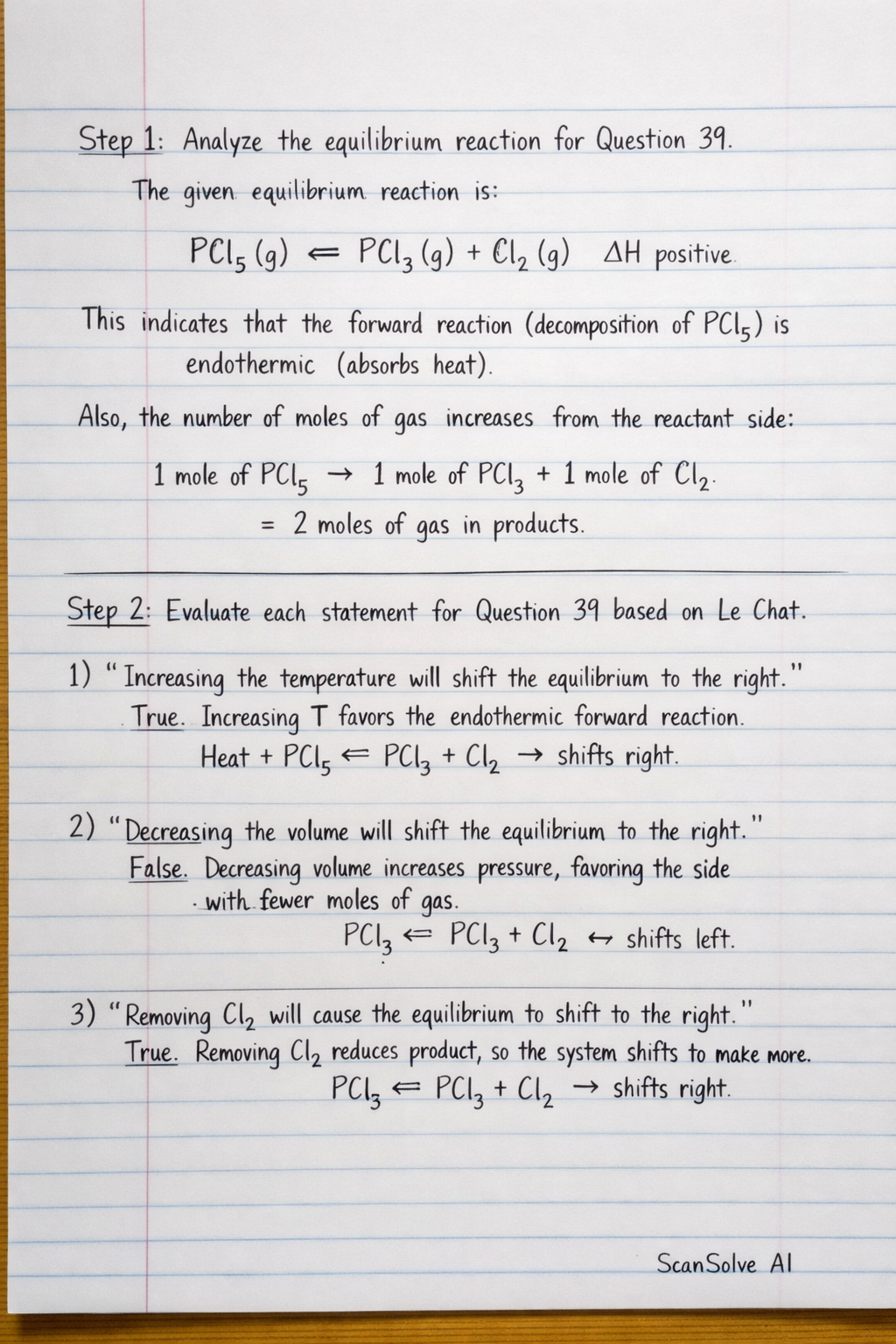
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Analyze the equilibrium reaction for Question 39. The given equilibrium reaction is: PCl_5(g) PCl_3(g) + Cl_2(g) H positive This indicates that the forward reaction (decomposition of PCl_5) is endothermic (absorbs heat).
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.