Step-by-step AI solutions. Ask your own question →

A functional group is an atom or a group of atoms responsible for the characteristic chemical properties of an organic c…
View Solution
But-1-ene is an alkene, which contains a carbon-carbon double bond. Alkenes undergo addition polymerisation.
View Solution
Hess's Law states that the total enthalpy change for a chemical reaction is independent of the pathway taken, as long as…
View Solution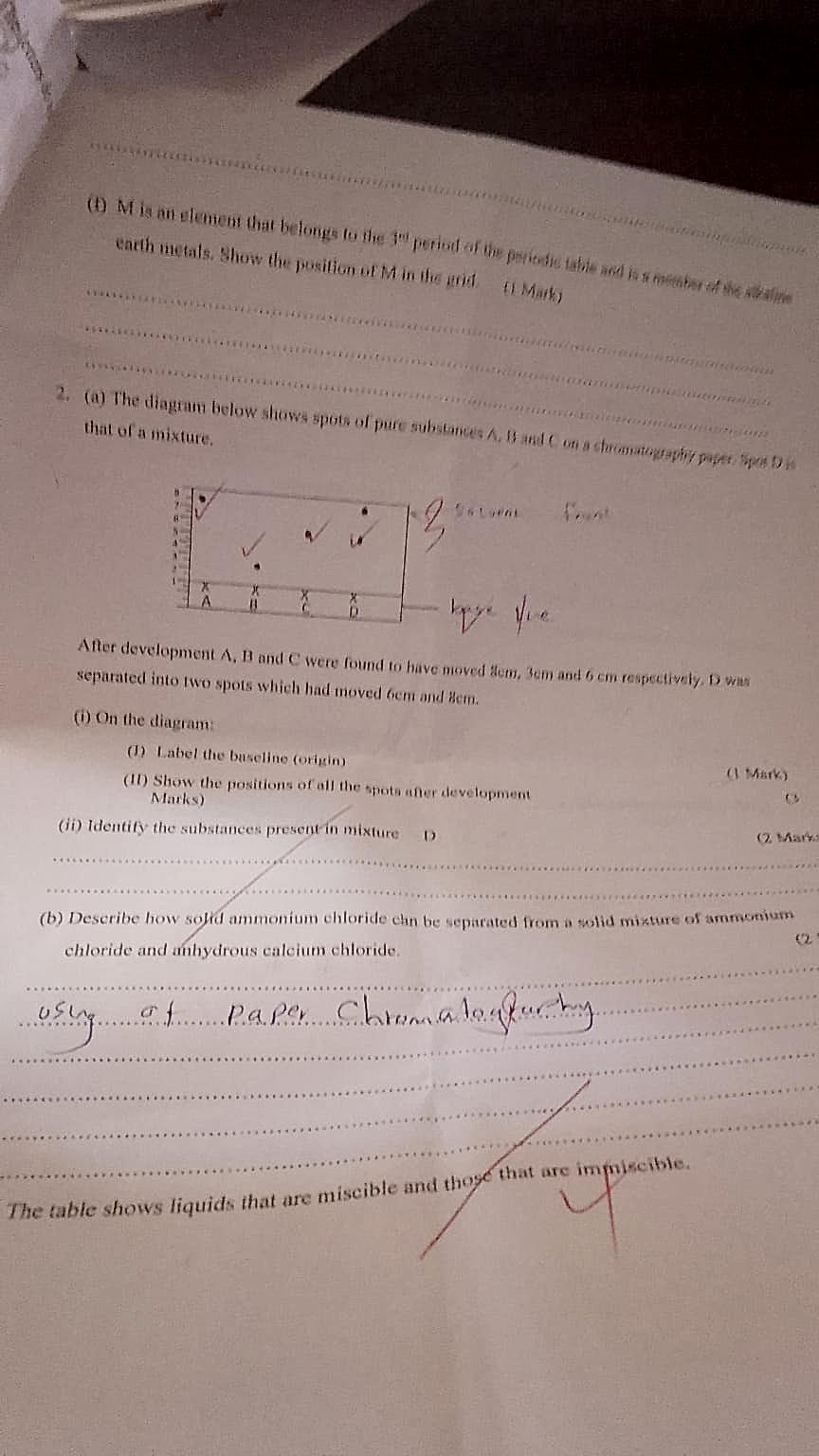
1 (b) M is an element that belongs to the 3rd period of the periodic table and is a member of the alkaline earth metals.…
View Solution
Define Relative Volatility and derive the expression XA = YA/α - (α - 1)YA from the ratio of partial pressure to mole fr…
View Solution17, 2. 35, 3. Chlorine, 4. 37 would be it mass number and we would call it ¹⁷Cl²⁰
View Solution
Dry Carbon (II) Oxide gas reacts with heated Lead (II) as shown in the equation below. PbO(s) + CO(g) → CO2(g) + Pb(s) a…
View Solution
Explain alkanoates. 2. State any three members of the alkanoate family. 3. Explain esterification. 4. Write a typical eq…
View Solution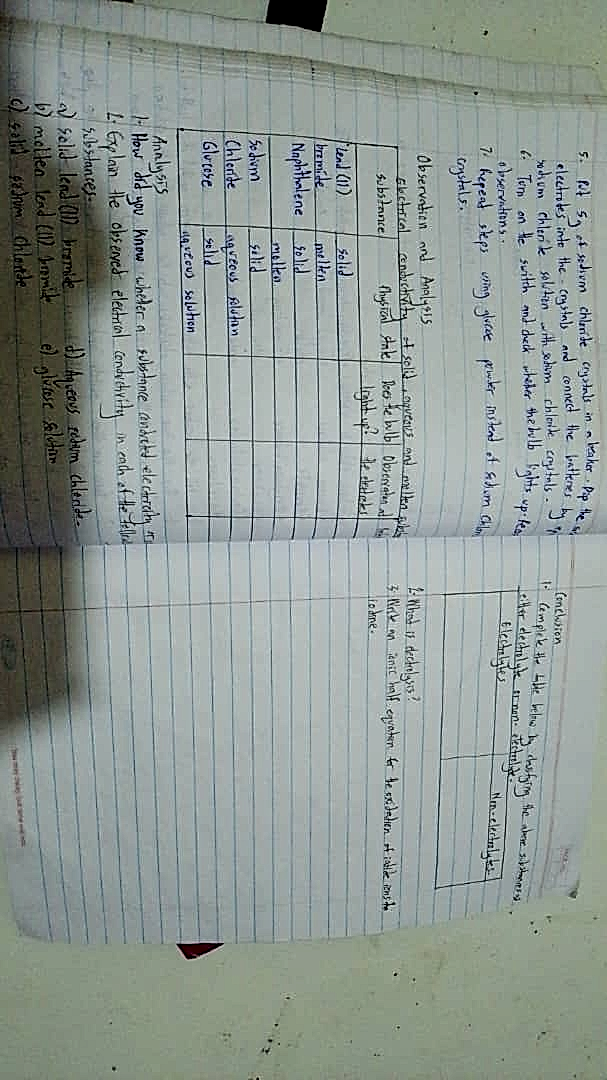
How did you know whether a substance conducted electricity in each of the following cases? a) solid lead (II) bromide b)…
View Solution
Organic Chemistry is often considered the most common branch due to the vast number of carbon-containing compounds and i…
View SolutionThe number of atoms of chlorine present in 5.85g of NaCl is (Na=23,Cl=35.5) 2. 2.25g of an oxide of copper on reduction…
View Solution
The chemical equation for the reaction between wood charcoal (carbon) and concentrated sulfuric (VI) acid is:
View SolutionChemistry is the study of matter — its composition, structure, properties, and the transformations it undergoes. From balancing equations to predicting reaction products, chemistry connects the microscopic world of atoms and molecules to the macroscopic phenomena you observe in the lab and in everyday life. A solid grasp of chemistry opens doors to careers in medicine, engineering, environmental science, and materials research.
Stoichiometry
Mole conversions, limiting reagents, percent yield, and balancing chemical equations.
Atomic Structure
Electron configurations, quantum numbers, periodic trends, and the relationship between structure and properties.
Chemical Bonding
Ionic, covalent, and metallic bonds, Lewis structures, VSEPR theory, and molecular geometry.
Acid-Base Chemistry
pH calculations, strong vs. weak acids, buffer solutions, and titration curves.
Thermochemistry
Enthalpy, Hess's law, calorimetry, bond energies, and spontaneity with Gibbs free energy.
Equilibrium
Equilibrium constants, Le Chatelier's principle, ICE tables, and solubility product calculations.
Redox Reactions
Oxidation states, half-reactions, balancing redox equations, electrochemical cells, and Nernst equation.
Organic Chemistry
Functional groups, naming conventions, reaction mechanisms, and stereochemistry fundamentals.
Gas Laws
Ideal gas law, partial pressures, molar volume, and real gas deviations.
Kinetics
Reaction rates, rate laws, activation energy, catalysts, and the Arrhenius equation.
ScanSolve analyzes your chemistry problem, identifies the concept area, and delivers a solution with every calculation shown and every reasoning step explained. For stoichiometry, it walks through mole ratios, shows unit conversions, and identifies the limiting reagent before calculating yield.
For equilibrium and acid-base problems, the solver sets up ICE tables, writes equilibrium expressions, and solves for unknowns — explaining assumptions like the small-x approximation and when they are valid. For organic chemistry, it traces reaction mechanisms step by step, showing electron movement and intermediate structures.
Each solution is designed to teach the underlying method so you can apply it to similar problems independently.
Master dimensional analysis early. Nearly every chemistry calculation involves unit conversions, and tracking units prevents most arithmetic errors.
Build a reaction type reference sheet: synthesis, decomposition, single replacement, double replacement, combustion. Recognizing the pattern instantly speeds up problem solving.
For organic chemistry, practice drawing mechanisms by hand. Understanding electron flow is the key to predicting products — memorizing reactions without mechanisms leads to confusion.
Use practice problems with answers to self-test regularly. Spaced repetition of problem types is more effective than marathon study sessions.
Send a photo of your chemistry homework and get step-by-step solutions instantly. No app download needed.
Message Us on WhatsAppCount atoms of each element on both sides, then adjust coefficients until both sides are equal. Start with the most complex molecule.
A mole is 6.022 × 10²³ particles (Avogadro’s number). It lets you convert between atoms/molecules and grams using molar mass.
Ionic bonds transfer electrons between a metal and nonmetal. Covalent bonds share electrons between nonmetals.
ScanSolve's AI solves chemistry problems step by step. Snap a photo of your homework or type your question to get instant, detailed solutions with clear explanations.
Yes, ScanSolve offers free chemistry homework help with step-by-step solutions. Upgrade to Plus for unlimited answers and priority support.
How to Balance Chemical Equations: Step-by-Step Method
Master balancing chemical equations with this step-by-step guide. Learn the trial and error method, tips for complex equations, and practice problems for chemistry students.
ChemistryThe Periodic Table Explained: Groups, Periods, Trends & Electron Configuration
Understand the periodic table including groups, periods, periodic trends, and electron configuration. A complete chemistry guide to elements and their properties.
BiologyPhotosynthesis Explained Step by Step: How Plants Make Food
Understand photosynthesis step by step. Learn the light reactions, Calvin cycle, and how plants convert sunlight into energy. Perfect for biology students.
BiologyCell Structure and Function: Organelles, Plant vs Animal Cells & Cell Membrane
Learn about cell structure and function including organelles, the cell membrane, and differences between plant and animal cells. Complete biology guide for students.
Get an instant AI-powered step-by-step solution in seconds.