This English question involves literary analysis, grammar, or writing skills. The detailed response below provides a well-structured answer with supporting evidence and clear explanations.
ScanSolve AI Answer
Answer
Here are the steps to solve the problem:
a) Plot a graph of mass (M/g) on the vertical axis and volume (V/cm) on the horizontal axis.
Step 1: Prepare the axes. Draw a horizontal axis for Volume (V/cm) ranging from 0 to at least 400 cm. Draw a vertical axis for Mass (M/g) ranging from 0 to at least 80 g.
Step 2: Plot the data points. Plot the following points: • • • • •
Step 3: Draw the line of best fit. Draw a straight line that passes through all the plotted points and extends to the origin .
b) (I) Determine the slope of the graph.
Step 1: Choose two points from the line. We can use any two points from the given data, for example, and .
Step 2: Calculate the slope. The slope is given by the formula: Substitute the chosen points: The slope of the graph is .
(II) State the significance of the slope. The slope of the mass versus volume graph represents the mass concentration (or density) of the sodium hydroxide solution. It indicates the mass of NaOH solute present per unit volume of the solution.
c) From the graph, determine the mass of the solute in of solution.
Step 1: Use the equation of the line. Since the graph is a straight line passing through the origin, its equation is , where is the slope. We found the slope .
Step 2: Calculate the mass for . The mass of the solute in of solution is .
d) Calculate the molar concentration of the sodium hydroxide in the standard solution. (Na=23, O=16, H=1)
Step 1: Calculate the molar mass of NaOH. Molar mass of NaOH = Atomic mass of Na + Atomic mass of O + Atomic mass of H Molar mass of NaOH = Molar mass of NaOH =
Step 2: Convert the mass concentration (slope) from g/cm to g/L. Mass concentration = Since : Mass concentration in g/L = Mass concentration in g/L =
Step 3: Calculate the molar concentration. Molar concentration = Molar concentration = Molar concentration = The molar concentration of the sodium hydroxide in the standard solution is .
e) State two precautions to be taken when preparing the standard solution. • Ensure the solute (NaOH) is pure and dry before weighing to avoid errors due to impurities or absorbed moisture. • Use a volumetric flask of the correct capacity and ensure the solution is made up to the mark accurately at the meniscus to ensure precise volume measurement.
3 done, 2 left today. You're making progress.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question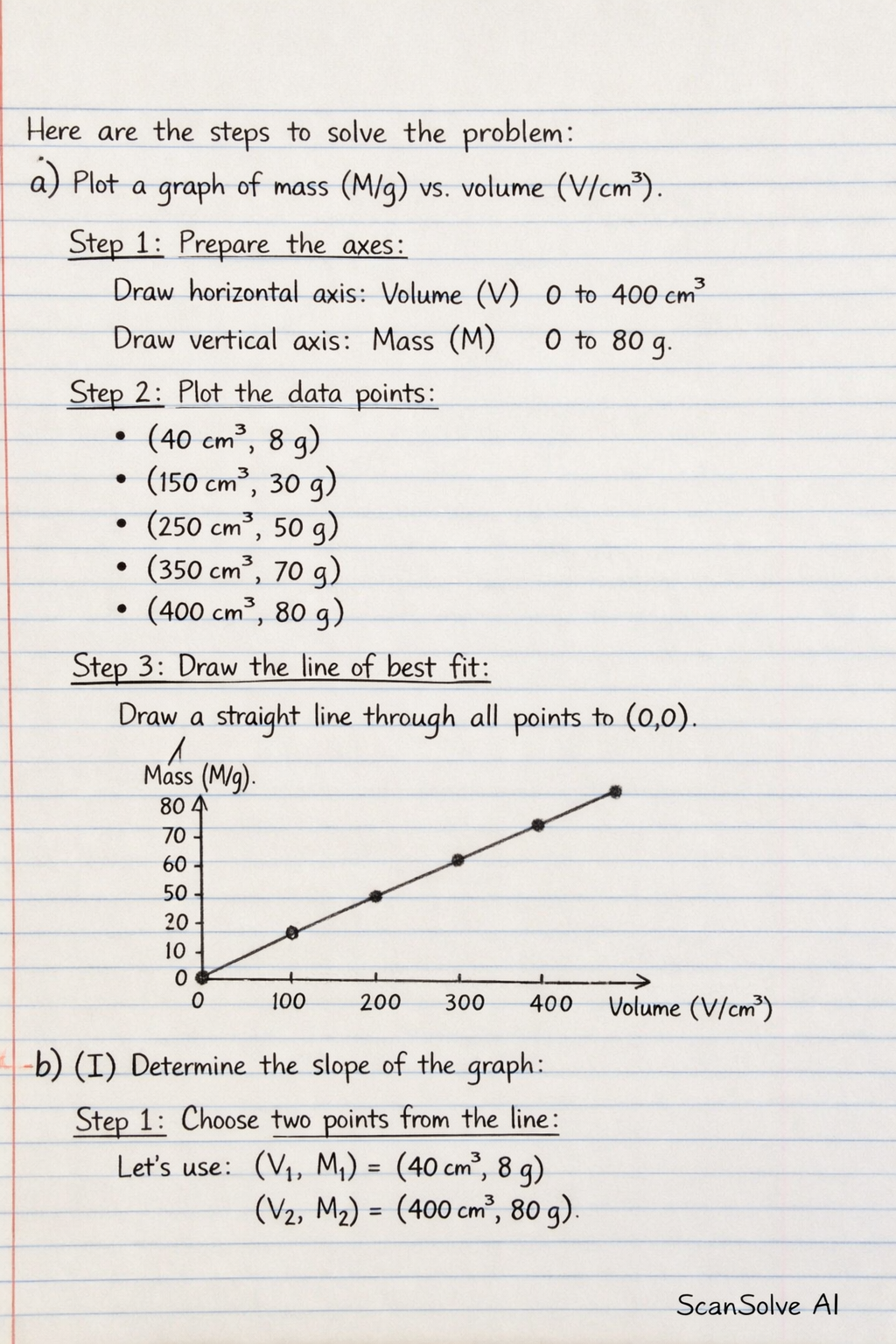
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
a) Plot a graph of mass (M/g) on the vertical axis and volume (V/cm^3) on the horizontal axis.
This English question involves literary analysis, grammar, or writing skills. The detailed response below provides a well-structured answer with supporting evidence and clear explanations.