This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.

ScanSolve AI Answer
Answer
-33.24 kJ
Here are the solutions to your questions:
1.1 The work done during adiabatic compression The process from point 1 to 2 is adiabatic compression (). Given: , , , .
Step 1: Calculate the pressure at point 2 () using the adiabatic relation.
Step 2: Calculate the work done during the adiabatic compression. The work done for a reversible adiabatic process is given by: The negative sign indicates work is done on the gas.
1.2 The work done during the constant pressure process The process from point 3 to 4 is a constant pressure process. Given: , , .
Step 1: Calculate the work done for the constant pressure process.
1.3 The volume at point 3 and point 5 in litres From the diagram: and . To convert from cubic meters to litres, multiply by 1000 ().
Step 1: Convert to litres.
Step 2: Convert to litres.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question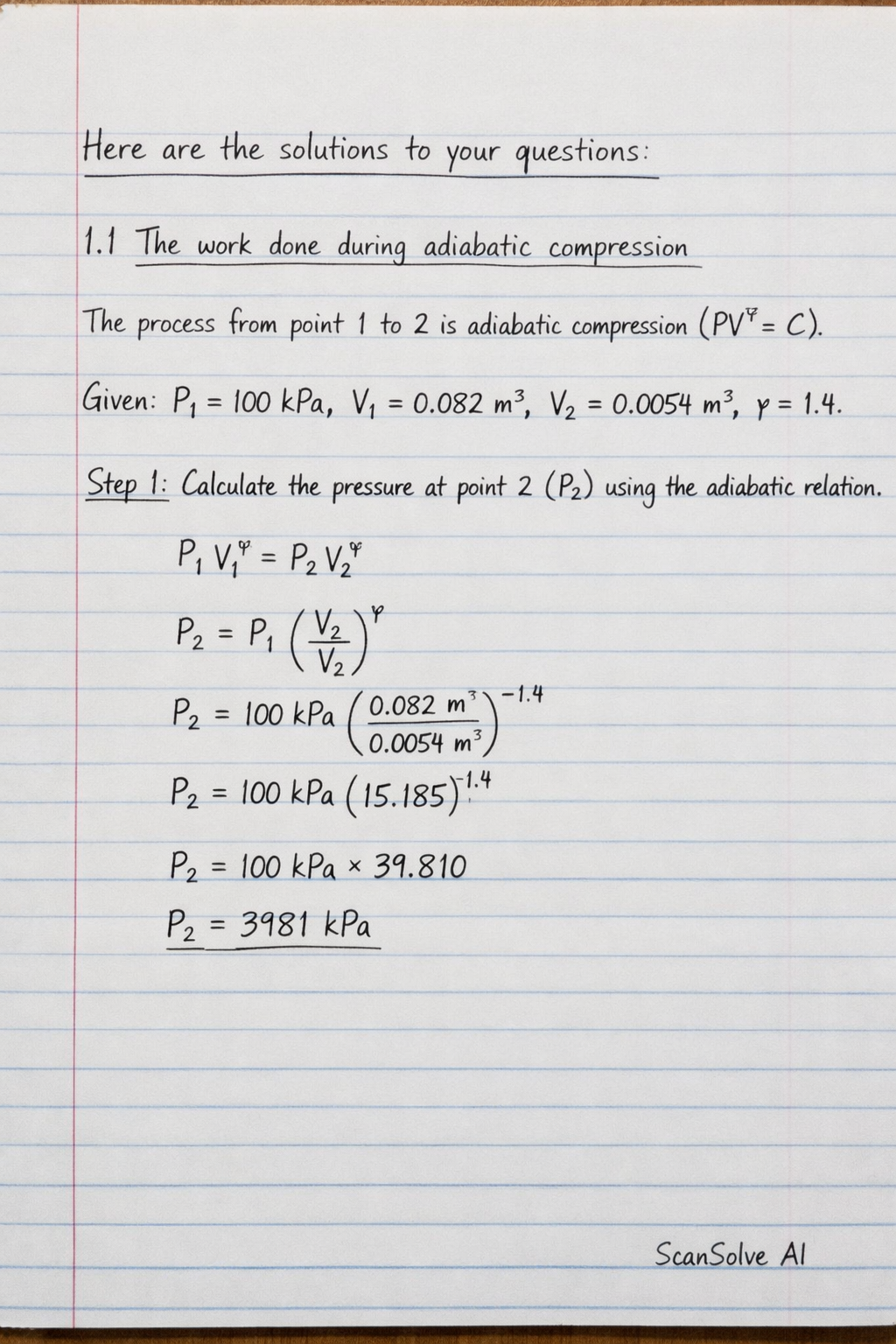
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
--- 1.1 The work done during adiabatic compression The process from point 1 to 2 is adiabatic compression (PV^ = C).
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.