This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.

ScanSolve AI Answer
Here's how to solve the problem using Hess's Law:
a) Draw an Energy cycle diagram linking the enthalpy of formation of ethanol with the enthalpy of combustion of its constituent elements.
The energy cycle diagram (Hess's Law cycle) for the formation of ethanol links the direct formation of ethanol from its elements to an alternative path involving the combustion of the elements and the combustion of ethanol.
The target reaction for the enthalpy of formation of ethanol () is: $2C(s) + 3H_2(g) + \frac{1}{2}O_2(g)
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question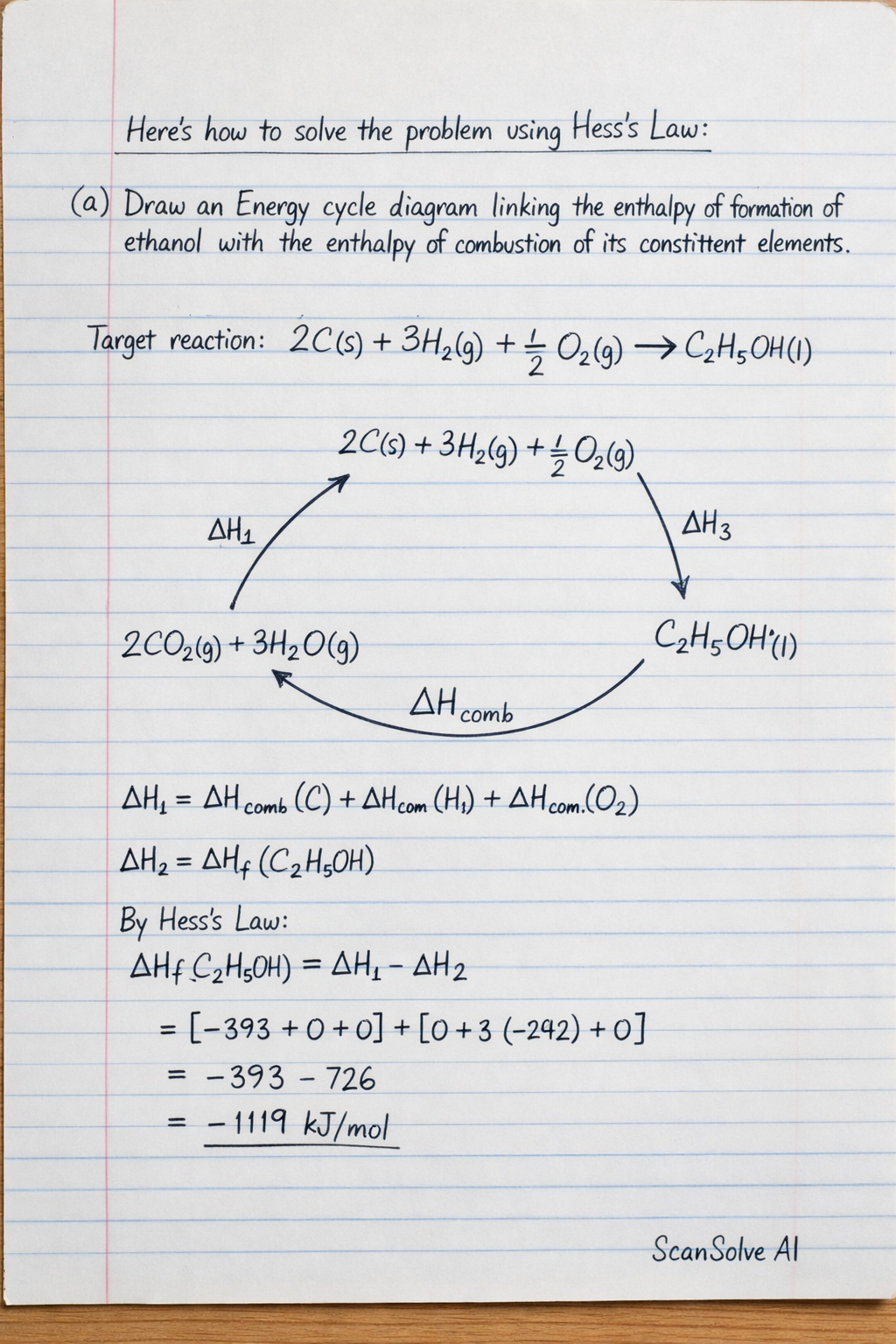
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Here's how to solve the problem using Hess's Law: a) Draw an Energy cycle diagram linking the enthalpy of formation of ethanol with the enthalpy of combustion of its constituent elements.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.