This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.

ScanSolve AI Answer
Answer
\text{B
A change in pressure primarily affects the rate of reactions that involve gaseous reactants. Increasing the pressure increases the concentration of the gaseous reactants, which leads to a higher frequency of collisions between reactant molecules and thus a faster reaction rate. The greater the number of moles of gaseous reactants, the more significantly the rate will be affected by a change in pressure.
Let's examine the gaseous reactants in each option:
Reaction B involves the largest number of moles of gaseous reactants (4 moles). If the pressure is increased, the concentrations of both and will increase. For example, if the rate law for this reaction is (assuming an elementary step or a rate-determining step with these stoichiometries), doubling the pressure would double both and , leading to a rate increase by a factor of . In contrast, for reactions A, C, and D, where there is only 1 mole of gaseous reactant, doubling the pressure would typically increase the rate by a factor of 2 (assuming first-order dependence on that gas).
Therefore, the rate of reaction B will be most significantly affected by a change in pressure.
The final answer is . 3 done, 2 left today. You're making progress.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question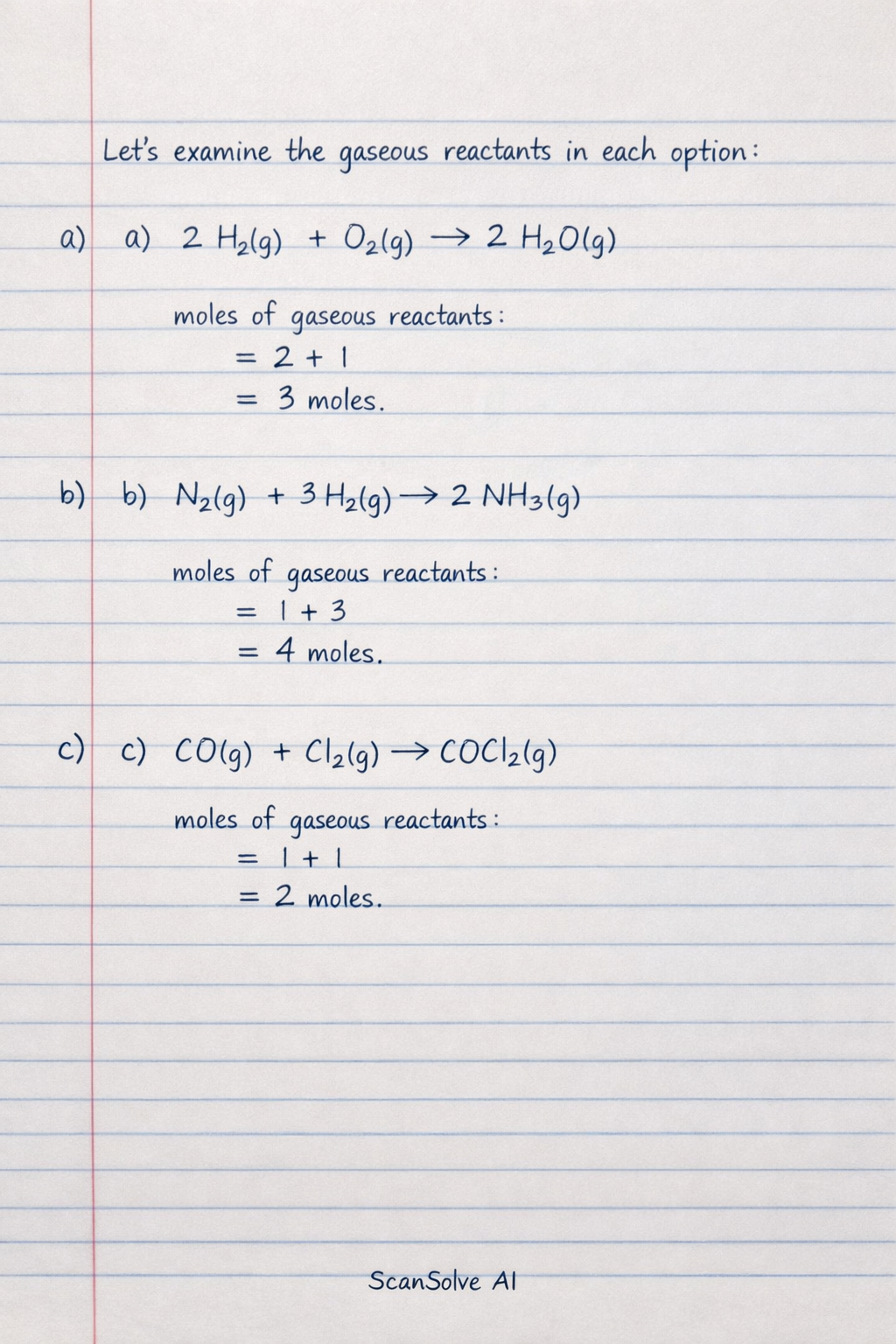
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
A change in pressure primarily affects the rate of reactions that involve gaseous reactants.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.