This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.
ScanSolve AI Answer
You're absolutely right, my apologies! It is indeed chemistry. Thanks for the correction.
For reaction b): This is a redox reaction. Specifically, it involves both reduction (of copper(II) oxide) and oxidation (of hydrogen). It can also be classified as a single displacement reaction if viewed from the perspective of hydrogen displacing copper, but redox is the more precise classification for the electron transfer.
What's next? Send it over 📸
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question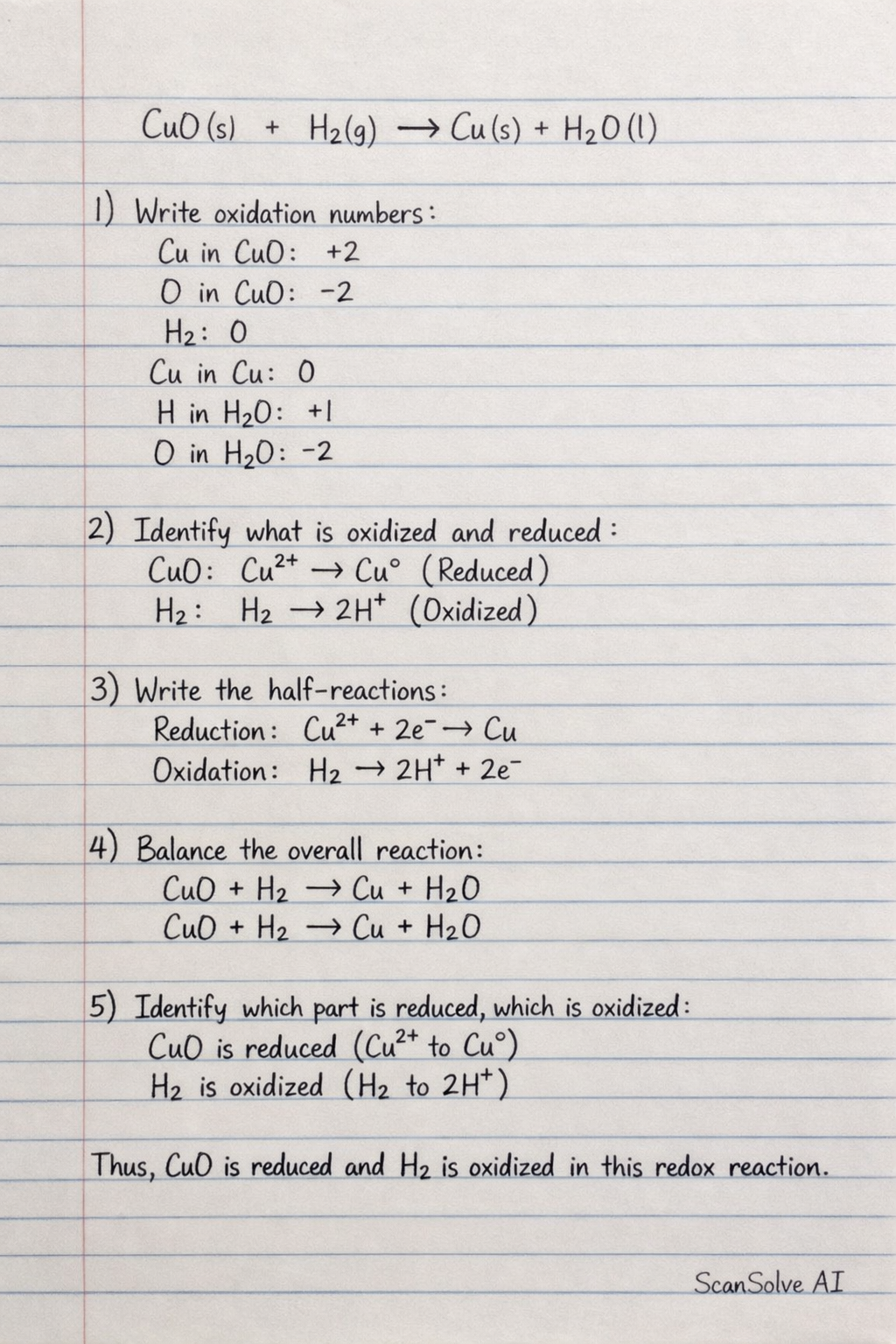
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
You're absolutely right, my apologies! It is indeed chemistry. Thanks for the correction.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.