This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.
ScanSolve AI Answer
Answer
3.21
Here's the solution for question 3:
3. The question asks for the pH of an HCl solution with a concentration of mol L.
Step 1: Determine the concentration of hydrogen ions. HCl is a strong acid, meaning it dissociates completely in water. Therefore, the concentration of H ions is equal to the concentration of HCl.
Step 2: Calculate the pH using the formula . Rounding to two decimal places, the pH is approximately 3.40.
Comparing this to the given options, (c) 3.21 is the closest value.
The correct option is (c) .
That's 2 down. 3 left today — send the next one.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question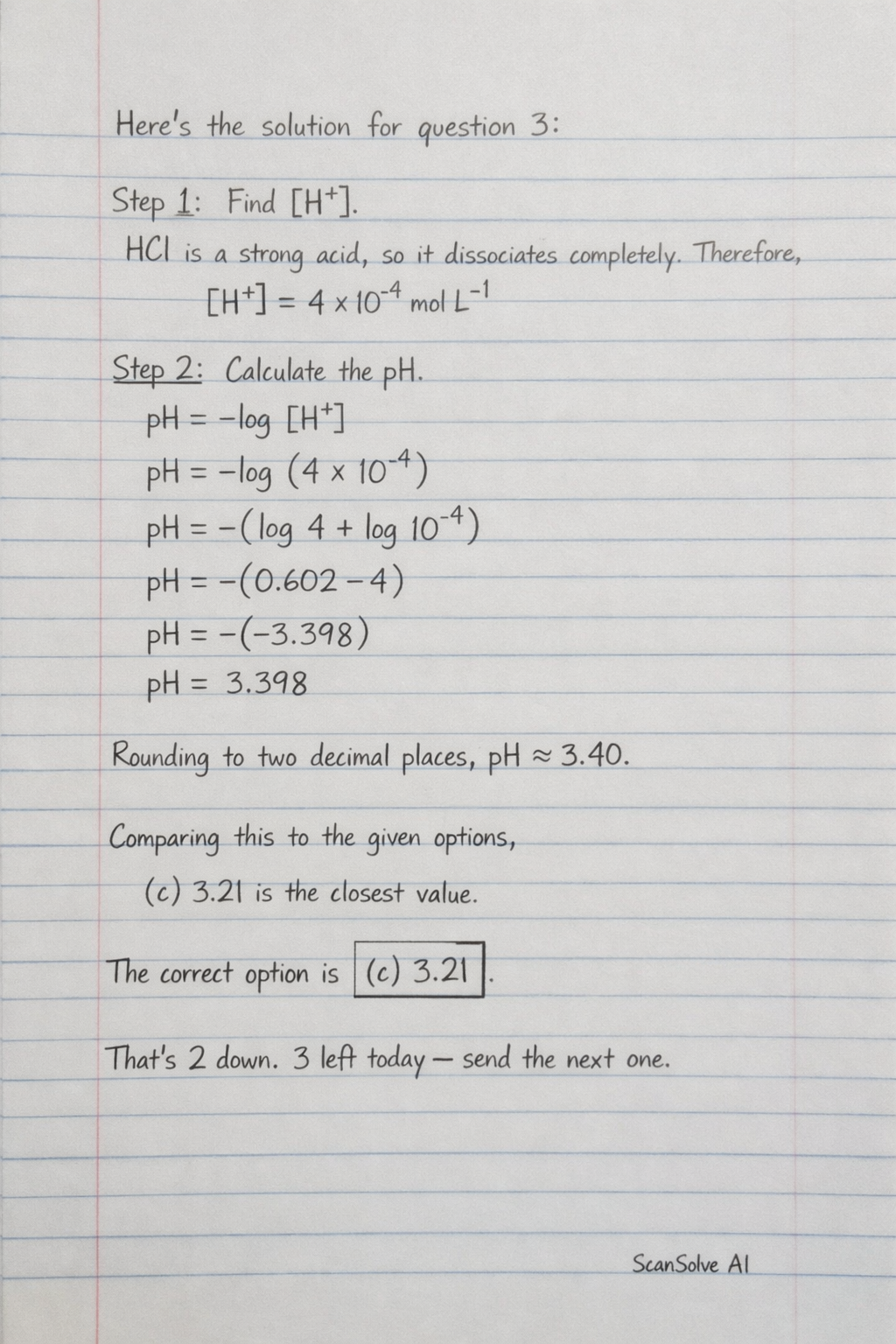
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Here's the solution for question 3: 3. The question asks for the pH of an HCl solution with a concentration of 4 × 10^-4 mol L^-1.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.