This geography problem is solved step by step below, with detailed explanations to help you understand the method and arrive at the correct answer.

ScanSolve AI Answer
Answer
A positive value for the enthalpy change () indicates that the reac
Here are the solutions to the questions:
65. When sodium dioxonitrate (III) () dissolves in water is positive, the process of dissolution is
66. The energy change () for the reaction is
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question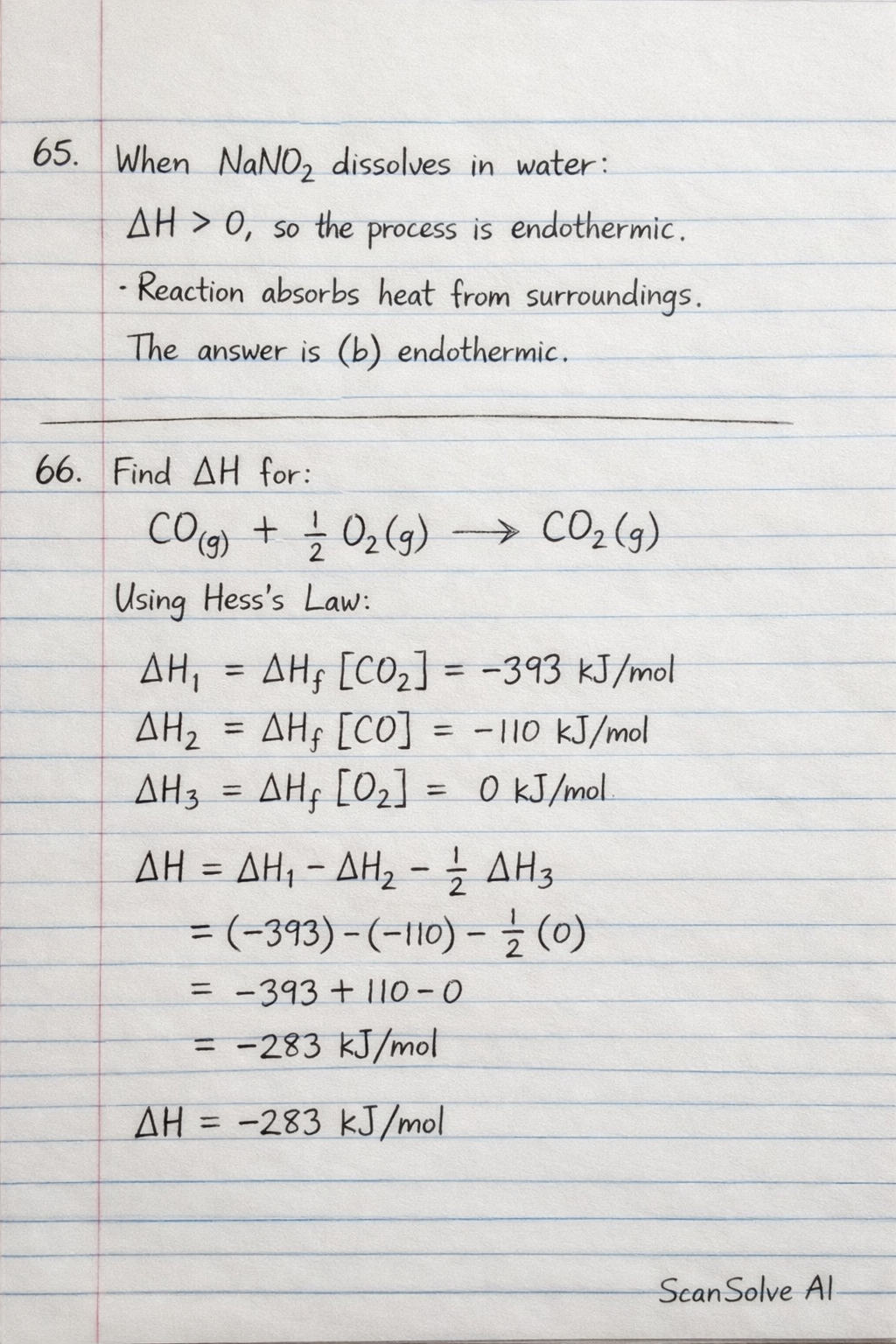
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
65. When sodium dioxonitrate (III) (NaNO_2) dissolves in water H is positive, the process of dissolution is A positive value for the enthalpy change ( H) indicates that the reaction absorbs heat from its surroundings.
This geography problem is solved step by step below, with detailed explanations to help you understand the method and arrive at the correct answer.