This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.

ScanSolve AI Answer
3 stepsAnswer
\text{1.04 mC
Here are the solutions to the physics problems.
1. Calculate the charge on the capacitor shown below.
Step 1: Calculate the equivalent capacitance of the two capacitors in series (, ).
Step 2: Calculate the total equivalent capacitance () by adding the series capacitance () to the parallel capacitor (). Convert to Farads:
Step 3: Calculate the total charge () using the total equivalent capacitance and the supply voltage (). The total charge on the capacitor combination is .
2. A motor powered by a 240V Main supply requires a current of 30A to lift a load of mass 3 Tonnes at the rate of 5M/Minute. Calculate the:
Given: Voltage () = 240 V Current () = 30 A Mass () = 3 Tonnes = Velocity () = 5 M/Minute = Acceleration due to gravity () =
a) Power input Step 1: Use the formula for electrical power input. The power input is .
b) Power output Step 1: Calculate the force required to lift the load (weight). Step 2: Calculate the power output using the force and velocity. The power output is .
c) Overall efficiency Step 1: Use the formula for efficiency. The overall efficiency is .
3.
a) State 3 factors that affect the rate of evaporation. • Temperature: A higher temperature increases the kinetic energy of liquid molecules, allowing more to escape the surface. • Surface area: A larger surface area exposes more liquid molecules to the air, increasing the rate of evaporation. • Air movement (wind): Wind carries away evaporated vapor molecules, reducing the concentration of vapor above the liquid and thus increasing the evaporation rate. • Humidity: Lower humidity (drier air) allows more water vapor to escape into the atmosphere.
b) State two differences and one similarity between evaporation and boiling.
Differences:
Similarity:
c) Calculate the quantity of heat required to change 0.50 kg of ice at -10°C completely into steam at 100°C.
Given: Mass () = 0.50 kg Specific heat capacity of ice () = 2100 J/kg/K Specific heat capacity of water () = 4200 J/kg/K Specific latent heat of fusion of ice () = J/kg Specific latent heat of vaporization of steam () = J/kg
The process involves four stages:
Step 1: Heat required to raise the temperature of ice from -10°C to 0°C ().
Step 2: Heat required to melt the ice at 0°C into water at 0°C ().
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question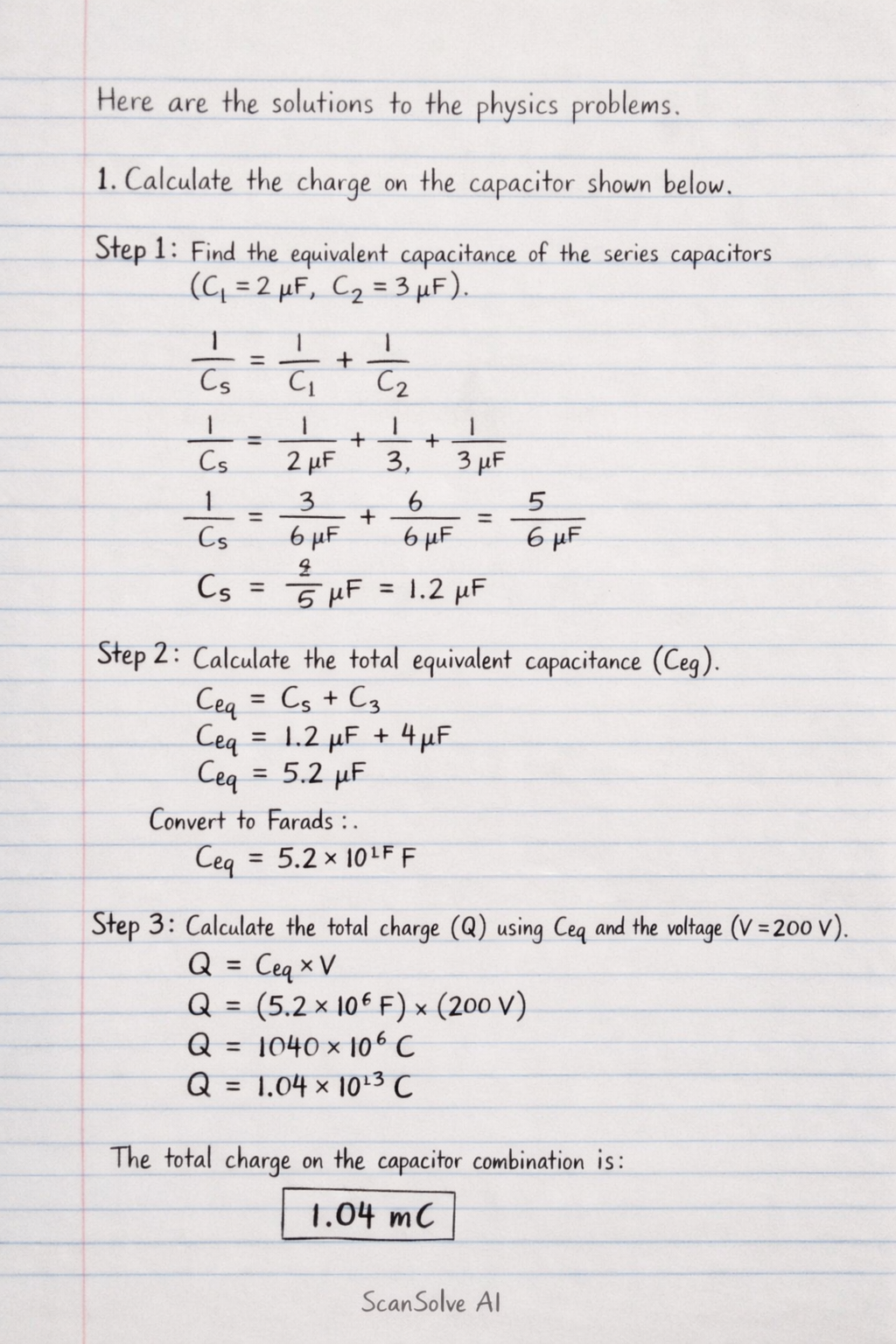
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Calculate the equivalent capacitance of the two capacitors in series (C_1 = 2 F, C_2 = 3 F).
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.