This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.

ScanSolve AI Answer
Answer
20 years
Step 1: Determine the number of half-lives () that have passed. The fraction of radioactive atoms remaining after time is given by the formula: where is the number of atoms remaining, is the initial number of atoms, and is the number of half-lives.
Given that the fraction of atoms left after 120 years is : We know that , so . Therefore, . This means 6 half-lives have passed in 120 years.
Step 2: Calculate the half-life () of the material. The number of half-lives () is also related to the total time () and the half-life () by the formula: We need to find , so we can rearrange the formula:
Substitute the given total time years and the calculated number of half-lives :
Step 3: Compare the result with the given options. The calculated half-life is 20 years, which corresponds to option C.
The half-life of the material is .
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question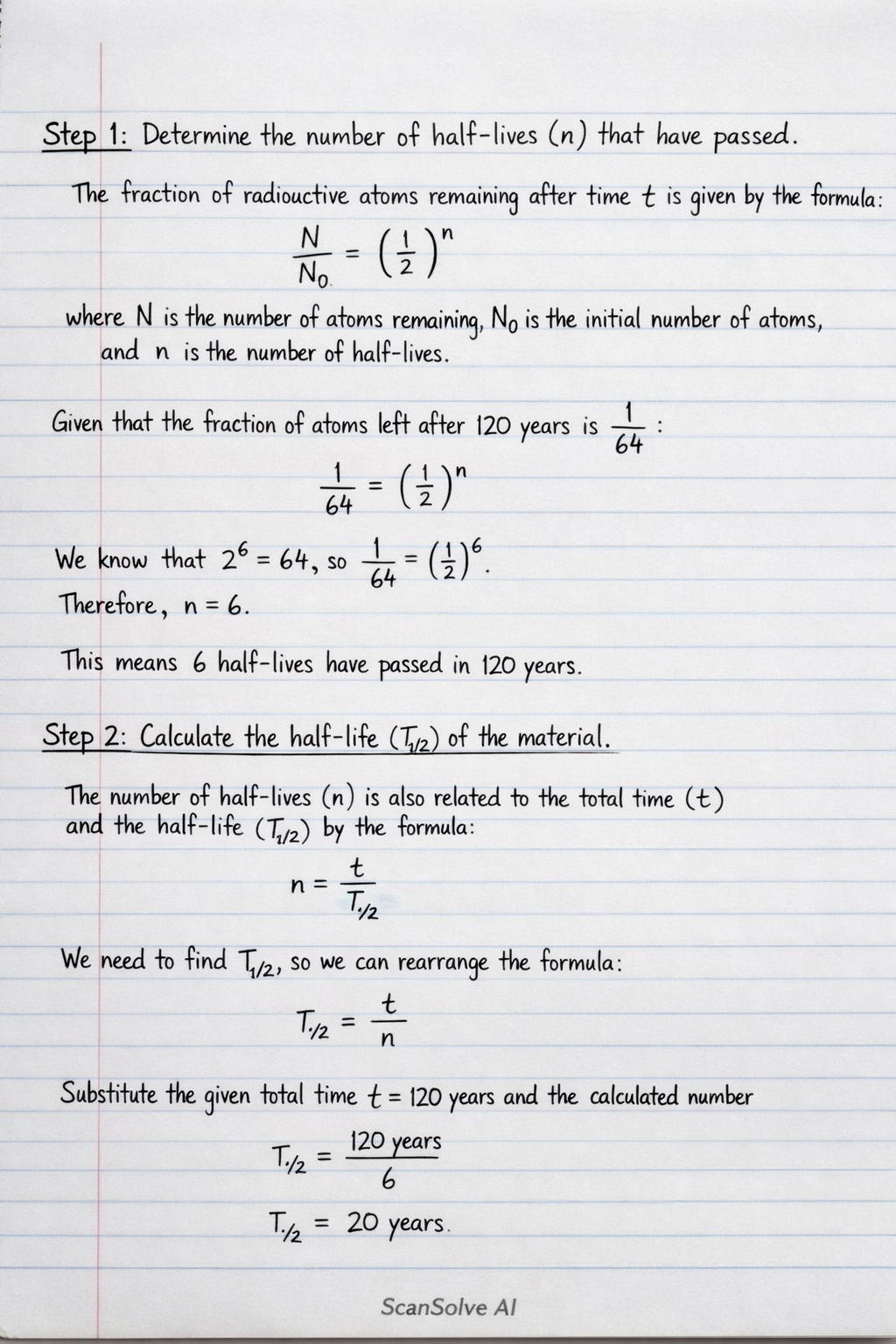
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Determine the number of half-lives (n) that have passed.
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.