This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.

ScanSolve AI Answer
This experiment aimed to determine the specific heat capacities of different metals by applying the principle of calorimetry and the law of conservation of energy. When a hot metal sample is placed into cooler water within an insulated calorimeter, heat transfers from the metal to the water until thermal equilibrium is reached. This heat transfer is quantified by the equation , where is the heat transferred, is the mass, is the specific heat capacity, and is the temperature change.
The core principle relies on the assumption that the heat lost by the metal sample is equal to the heat gained by the water. Therefore, . By measuring the masses and temperature changes of both the metal and water, and knowing the specific heat of water (), the specific heat of the metal () can be calculated.
Several potential sources of error could influence the accuracy of the determined specific heat values. Heat loss to the surroundings is a primary concern, as no calorimeter is perfectly insulated, meaning some heat from the metal might escape to the environment rather than being fully absorbed by the water. This would lead to an underestimation of the metal's specific heat. Incomplete thermal equilibrium between the metal and water, or delays in transferring the hot metal, could also result in inaccurate temperature measurements. Furthermore, the heat absorbed by the calorimeter itself was not explicitly included in the provided energy balance equations, which would cause an underestimation of the total heat gained by the system.
To enhance the experiment's precision, improvements could include using a more effectively insulated calorimeter to minimize heat exchange with the surroundings. It would also be beneficial to measure and incorporate the heat capacity of the calorimeter into the calculations to account for the heat absorbed by the calorimeter material. Ensuring rapid transfer of the metal sample and allowing ample time for the system to reach true thermal equilibrium would also improve the reliability of the temperature readings.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question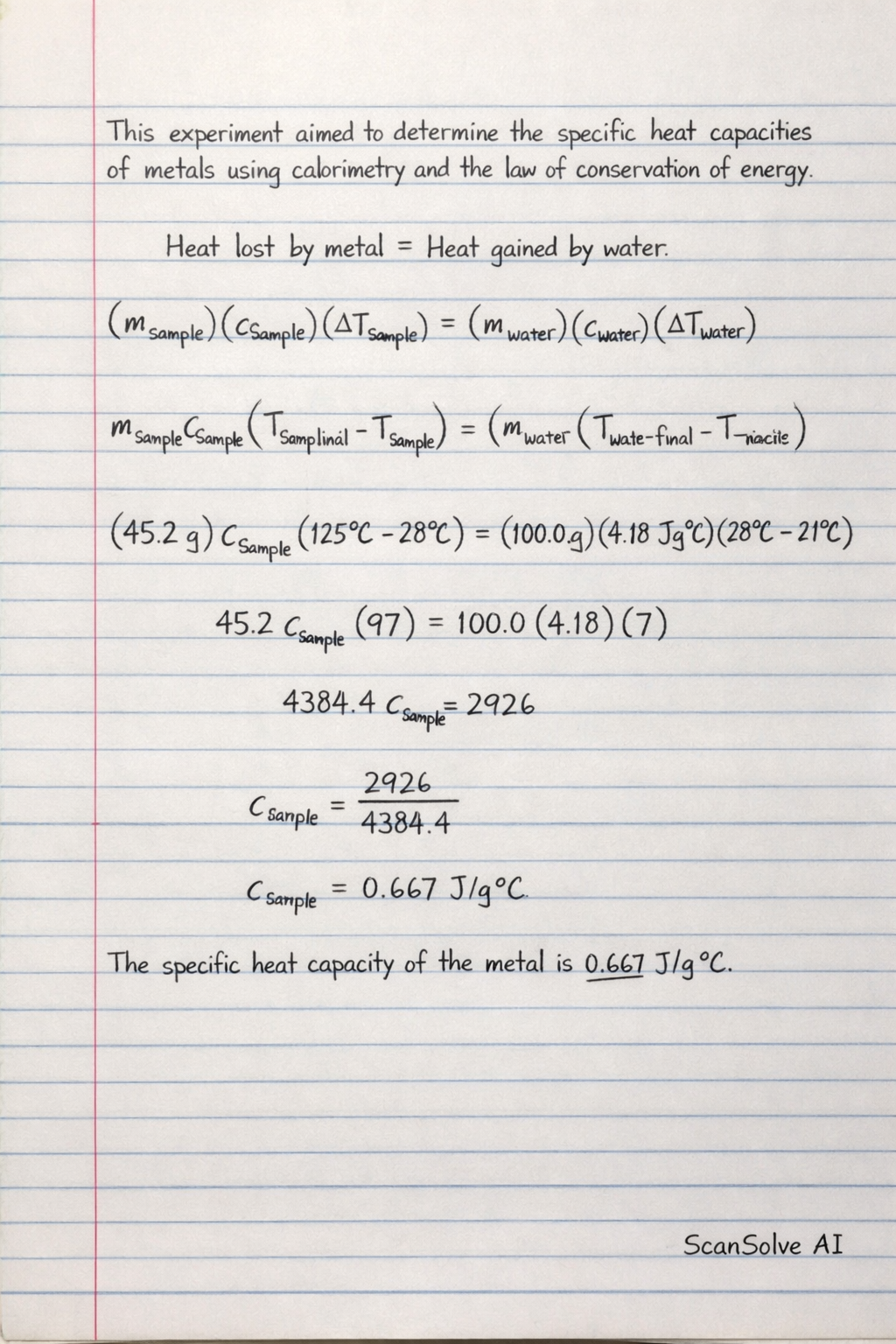
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
This experiment aimed to determine the specific heat capacities of different metals by applying the principle of calorimetry and the law of conservation of energy.
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.