This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.

ScanSolve AI Answer
You're on a roll — i) To determine the rate of reaction at 12 minutes: Step 1: Locate the point on the curve corresponding to minutes on the time (x-axis). Step 2: Draw a tangent to the curve at this specific point. Step 3: Calculate the gradient (slope) of this tangent. The gradient, , represents the rate of reaction at minutes.
ii) The production of oxygen in region is faster than in region . Explanation: In region , the concentration of reactants is higher, leading to a greater frequency of effective collisions between reactant particles and thus a faster rate of reaction. As the reaction proceeds into region , the concentration of reactants decreases, resulting in fewer effective collisions and a slower rate of oxygen production.
iii) The equation for the decomposition of hydrogen peroxide is:
c) i) A major source of nitrogen gas in the Haber process is air.
Got more? Send 'em!
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question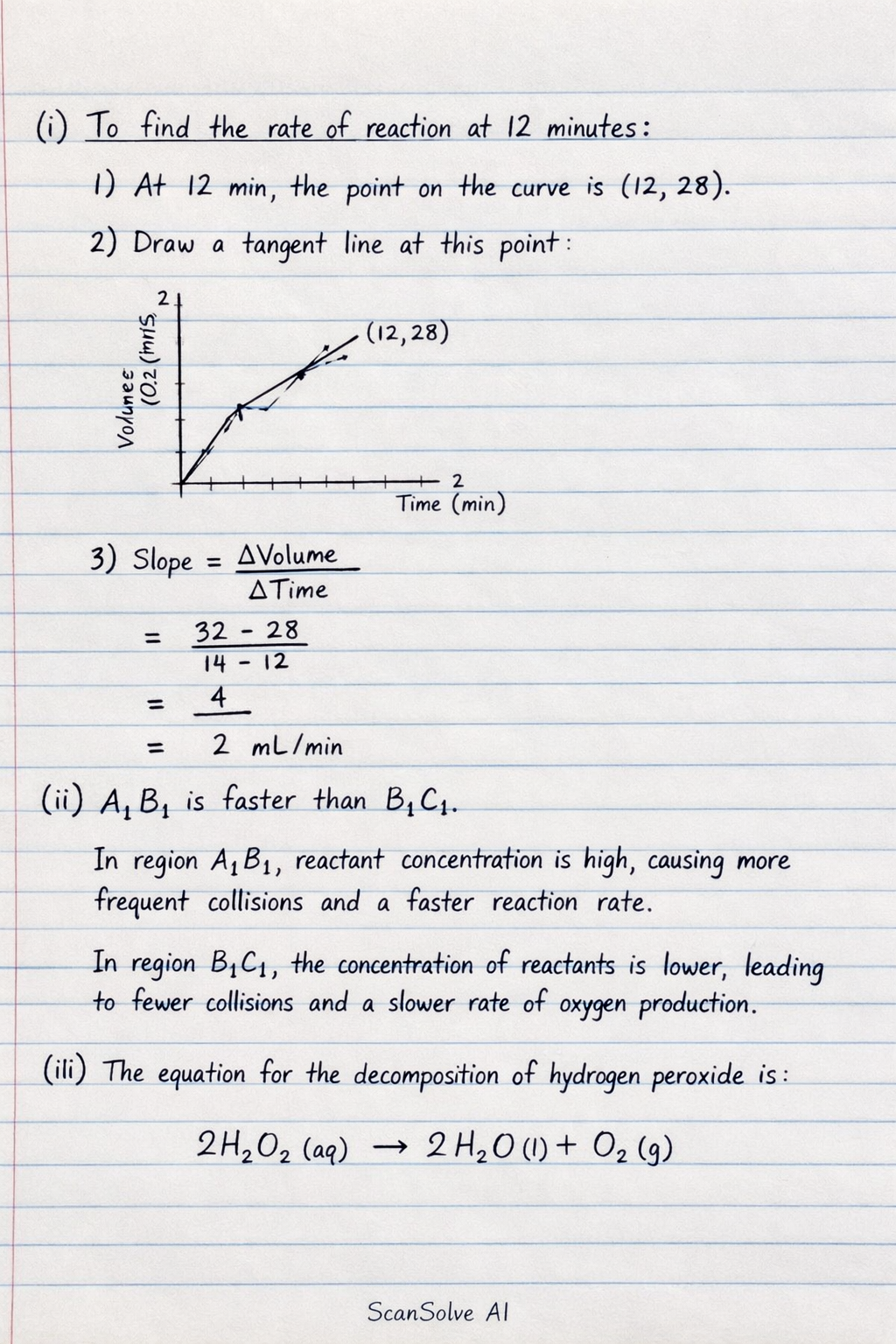
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
You're on a roll — i) To determine the rate of reaction at 12 minutes: Step 1: Locate the point on the curve corresponding to 12 minutes on the time (x-axis).
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.