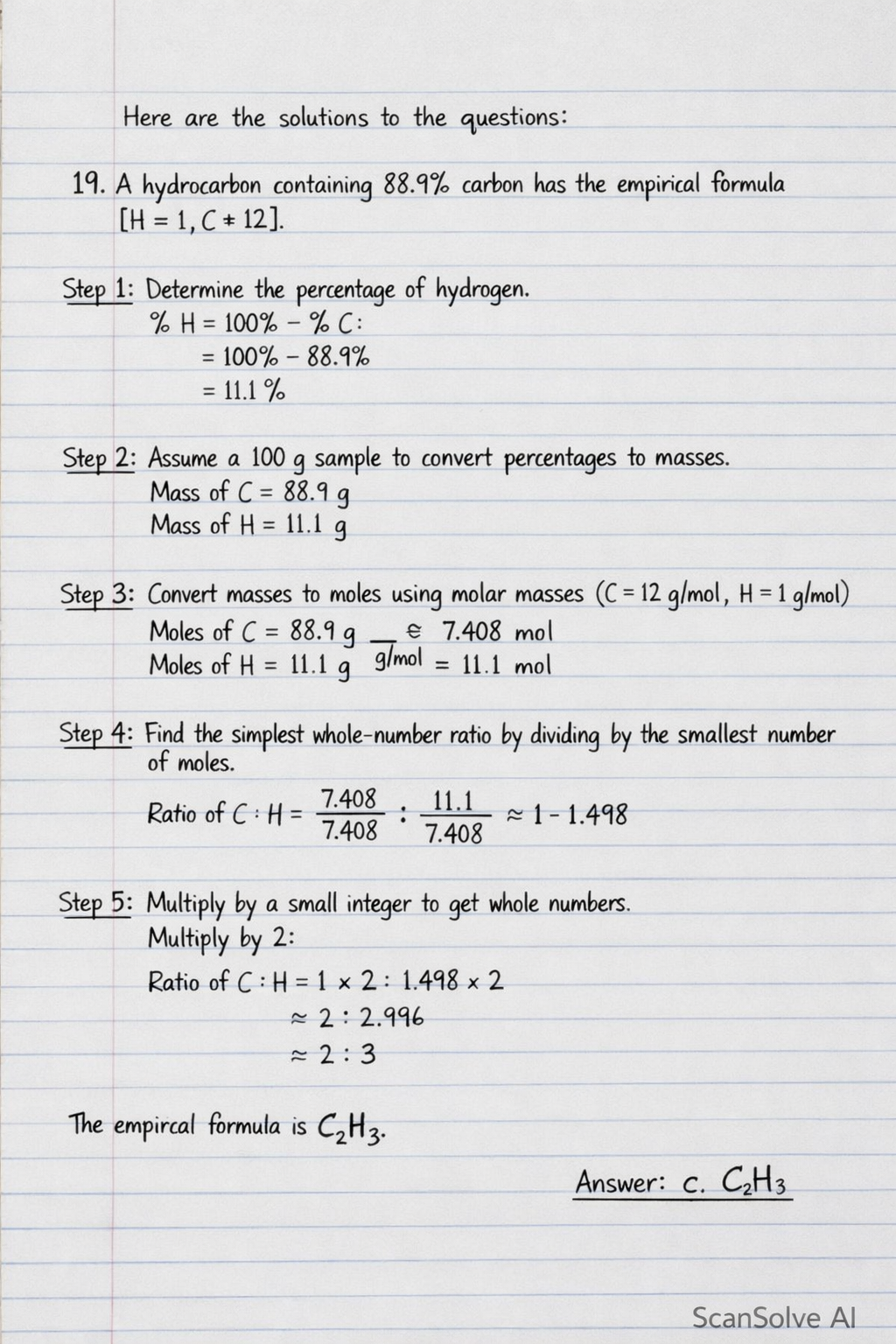Here are the solutions to the questions:
19. A hydrocarbon containing 88.9% carbon has the empirical formula [H=1, C=12].
Step 1: Determine the percentage of hydrogen.
Since it's a hydrocarbon, it contains only carbon and hydrogen.
%H=100%−%C=100%−88.9%=11.1%
Step 2: Assume a 100g sample to convert percentages to masses.
Mass of C = 88.9g
Mass of H = 11.1g
Step 3: Convert masses to moles using molar masses (C=12 g/mol, H=1 g/mol).
Moles of C = 12g/mol88.9g≈7.408mol
Moles of H = 1g/mol11.1g=11.1mol
Step 4: Find the simplest whole-number ratio by dividing by the smallest number of moles.
Ratio of C:H = 7.4087.408:7.40811.1≈1:1.498
Step 5: Multiply by a small integer to get whole numbers.
Multiply by 2:
Ratio of C:H = 1×2:1.498×2≈2:2.996≈2:3
The empirical formula is C₂H₃.
- Answer: c.C2H3
20. Which of the following methods can be used to separate blood cells from plasma?
- Explanation: Blood cells are suspended in plasma and are denser. Centrifugation uses centrifugal force to separate components of different densities, making it ideal for separating blood cells from plasma.
- Answer: a.centrifugation
21. The purity of a solid sample can best be determined by its...
- Explanation: The melting point of a pure solid is a specific, sharp temperature. Impurities typically lower the melting point and broaden the melting range, making it a reliable indicator of purity.
- Answer: b.meltingpoint
22. The most suitable process of obtaining water from an acre solution of sugar is...
- Explanation: To obtain pure water from a sugar solution, the water needs to be separated from the dissolved, non-volatile sugar. Distillation involves boiling the solution to vaporize the water, then condensing the vapor back into pure liquid water, leaving the sugar behind.
- Answer: b.distillation
23. Potassium trioxonitrate(V) can be obtained from its solution by...
- Explanation: Potassium trioxonitrate(V) (KNO₃) is a soluble salt. To obtain the solid salt from its solution, crystallization is the most suitable method. This involves evaporating the solvent until the solution is saturated, then cooling it to allow the salt to crystallize out.
- Answer: c.crystallization
24. A mixture containing two salts of different solubilities can be separated by a. chromatography b. distillation c. crystallization d. evaporation
- Explanation: When two salts have different solubilities in a solvent, they can be separated by fractional crystallization. This involves dissolving the mixture, then carefully changing conditions (e.g., cooling) to cause one salt to crystallize out before the other. Option c, crystallization, is the general principle.
- Answer: c.crystallization
25. The separation technique that is suitable for separating iodine from tetrachloromethane is a. solvent extraction b. fractional distillation c. decantation d. filtration
- Explanation: Iodine is a solid that dissolves in tetrachloromethane (CCl₄), which is a liquid. To separate a dissolved solid from a liquid solvent, fractional distillation is suitable. CCl₄ has a boiling point of 76.7°C, while iodine sublimes at 113.7°C (and melts at the same temperature). The CCl₄ would distill off, leaving the solid iodine behind.
- Answer: b.fractionaldistillation
26. When an ionic bond is broken, bonding electrons are...
- Explanation: An ionic bond forms when electrons are completely transferred from a metal (electropositive atom) to a non-metal (electronegative atom). When the bond breaks, the electrons remain with the atom that originally gained them, which is the most electronegative atom.
- Answer: c.gainedbythemostelectronegativeatom
27. The following molecules have double covalent bonds between two atoms except...
- Explanation:
- a. Oxygen (O₂): Has a double bond (O=O).
- b. Carbon(IV) oxide (CO₂): Has two double bonds (O=C=O).
- c. Ethene (C₂H₄): Has one double bond (H₂C=CH₂).
- d. Water (H₂O): Has two single bonds (H-O-H).
- Answer: d.water
28. The formation of a bond between hydrogen and a highly electronegative atom results in a...
- Explanation: When hydrogen bonds with a highly electronegative atom (e.g., O, N, F), the electron pair in the bond is pulled more towards the electronegative atom. This creates an uneven distribution of charge, leading to a polar covalent bond. The property of having these separated partial charges is called polarity.
- Answer: a.polarity
29. The molecule that has a non-polar covalent bond is a. H₂O b. HCl c. NH₃ d. Cl₂
- Explanation: A non-polar covalent bond forms between two atoms with identical or very similar electronegativity, resulting in equal sharing of electrons.
- a. H₂O: O-H bonds are polar.
- b. HCl: H-Cl bond is polar