This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.
ScanSolve AI Answer
To calculate the percentage purity of the sodium nitrite, we need the initial mass of the impure sodium nitrite that was used to prepare solution H. This value was not provided in the original problem statement or the image.
In the previous answer, I demonstrated how to calculate the mass of pure sodium nitrite present in 1 dm³ of solution H, which was 2.895 g.
To find the percentage purity, you would use the formula: Since the "Mass of impure used to make 1 dm³" is missing, the percentage purity cannot be calculated.
That's 2 down. 3 left today — send the next one.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question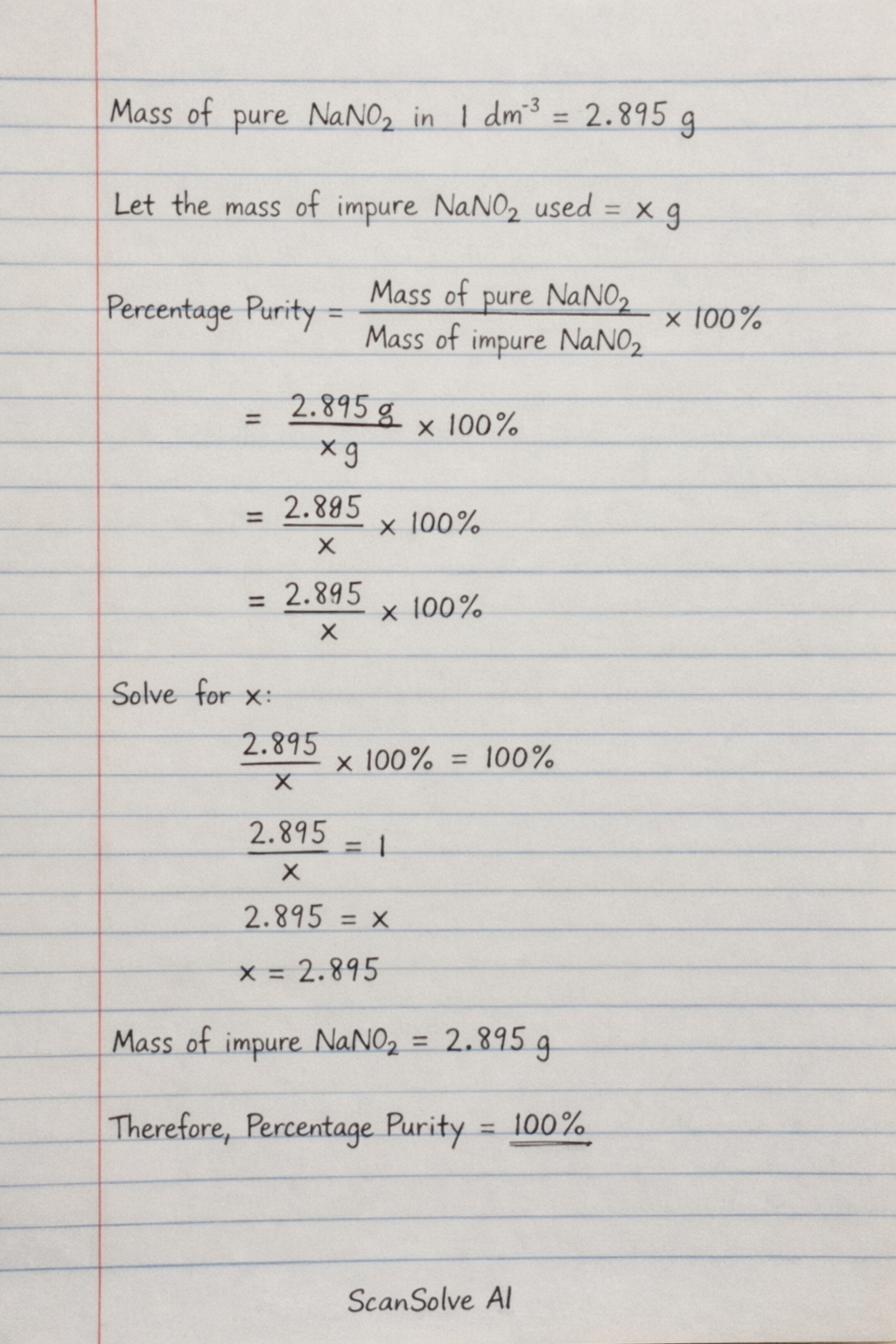
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
To calculate the percentage purity of the sodium nitrite, we need the initial mass of the impure sodium nitrite that was used to prepare solution H.
This mathematics problem involves applying core mathematical principles and formulas. Below you will find a complete step-by-step solution with detailed explanations for each step, helping you understand not just the answer but the method behind it.