This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.

ScanSolve AI Answer
Answer
To calculate the frequency of the photon, we use the relationship between energy and frequency for a photon, .
Step 1: Convert the given energy from electron volts (eV) to Joules (J). The energy () is . We know that .
Step 2: Use Planck's equation to find the frequency (). The equation is , where is Planck's constant (). Rearrange the formula to solve for :
Rounding to three significant figures:
The frequency of the photon is:
Got more? Send 'em.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question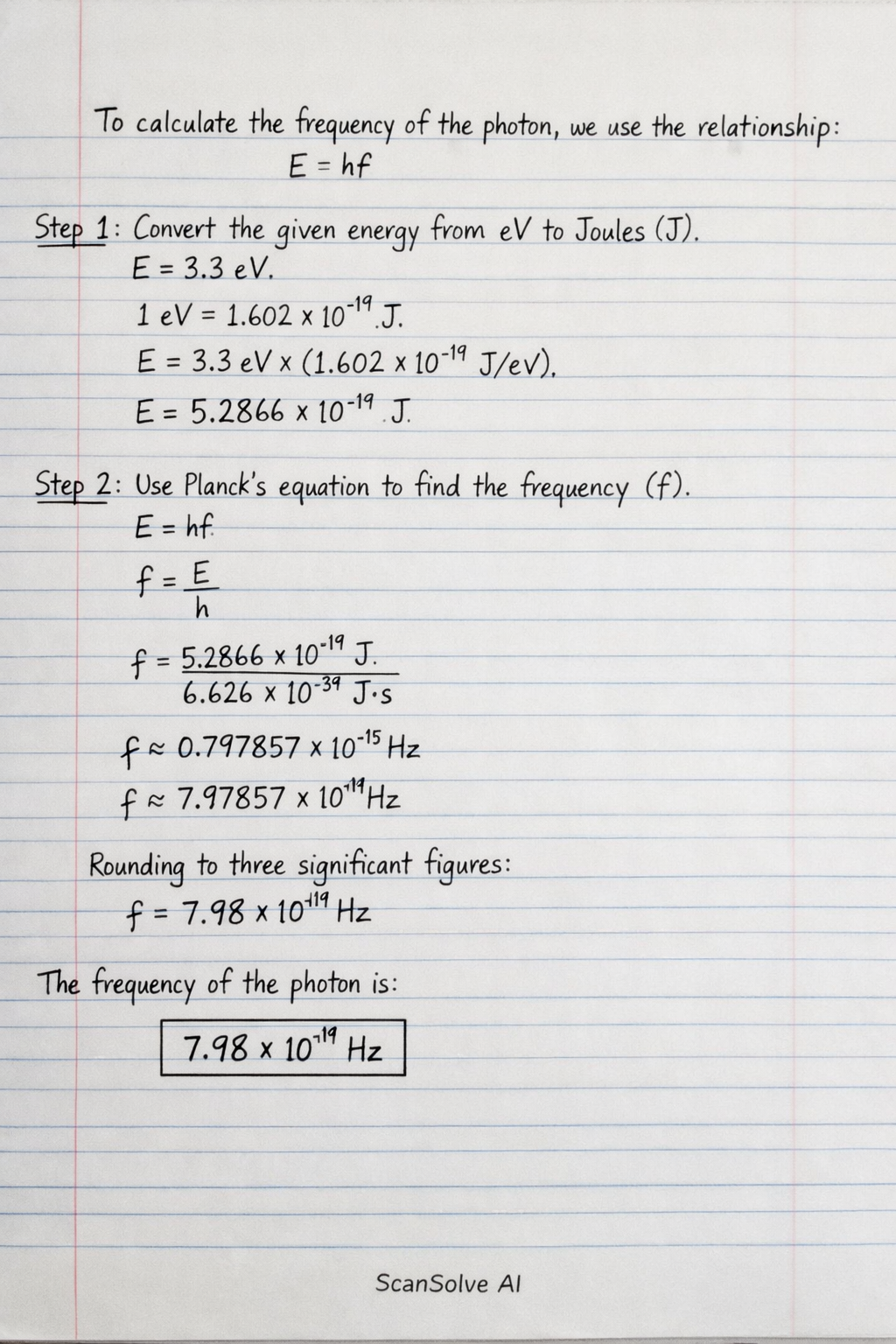
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
To calculate the frequency of the photon, we use the relationship between energy and frequency for a photon, E = hf.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.