This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.
ScanSolve AI Answer
A single excited hydrogen atom, at any given instant, will emit one wavelength.
Here's why:
The full hydrogen emission spectrum, which shows multiple distinct wavelengths (lines), is observed from a macroscopic sample of hydrogen gas. In such a sample, many atoms are simultaneously undergoing various different possible electron transitions, each contributing a photon of a specific wavelength to the overall observed spectrum.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question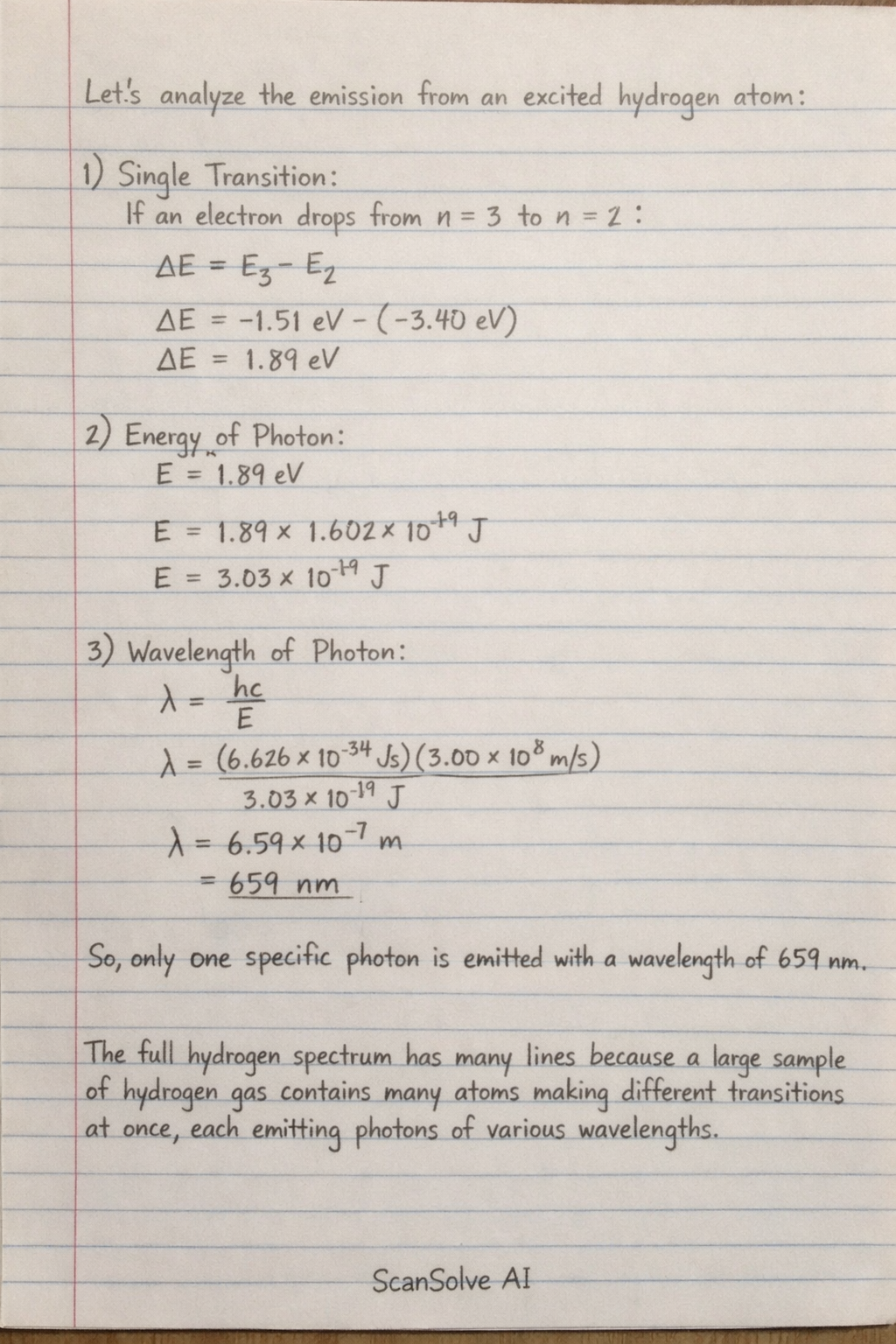
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
A single excited hydrogen atom, at any given instant, will emit one wavelength. Here's why: Single Transition: When an electron in a single hydrogen atom transitions from a higher energy level to a lower one, it undergoes one specific energy change*.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.