This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
Q9. A compound contains 46.7% of silicon (Si) and 53.3% oxygen (O) by mass. Find the empirical formula of the compound, given the relative atomic mass of silicon (Si) = 28 and oxygen (O) = 16.
Step 1: Assume a 100 g sample. This means we have 46.7 g of Si and 53.3 g of O.
Step 2: Convert mass to moles for each element.
Step 3: Find the simplest whole number ratio of moles. Divide each number of moles by the smallest number of moles (1.6679 mol).
The simplest whole number ratio of Si:O is 1:2.
Step 4: Write the empirical formula. The empirical formula is .
Q10. Caffeine is a compound found in coffee and tea. The percentage composition of caffeine is 49.1% carbon, 5.1% hydrogen, 16.5% oxygen and 28.9% nitrogen. The relative molecular mass of caffeine is 195. Find the molecular formula of caffeine, given the atomic mass of carbon (C) = 12, hydrogen (H) = 1, oxygen (O) = 16, and nitrogen (N) = 14.
Step 1: Assume a 100 g sample and convert percentages to masses.
Step 2: Convert mass to moles for each element.
Step 3: Find the simplest whole number ratio of moles. Divide each number of moles by the smallest number of moles (1.0313 mol for O).
The simplest whole number ratio of C:H:O:N is 4:5:1:2.
Step 4: Write the empirical formula. The empirical formula is .
Step 5: Calculate the empirical formula mass.
Step 6: Determine the factor 'n'. Given relative molecular mass = 195.
Step 7: Write the molecular formula. Multiply the subscripts in the empirical formula by n=2.
Molecular formula = (C_4H_5ON_2)_2 = \text{C_8H_{10}O_2N_4}Get instant step-by-step solutions to any question. Free to start.
Ask Your Question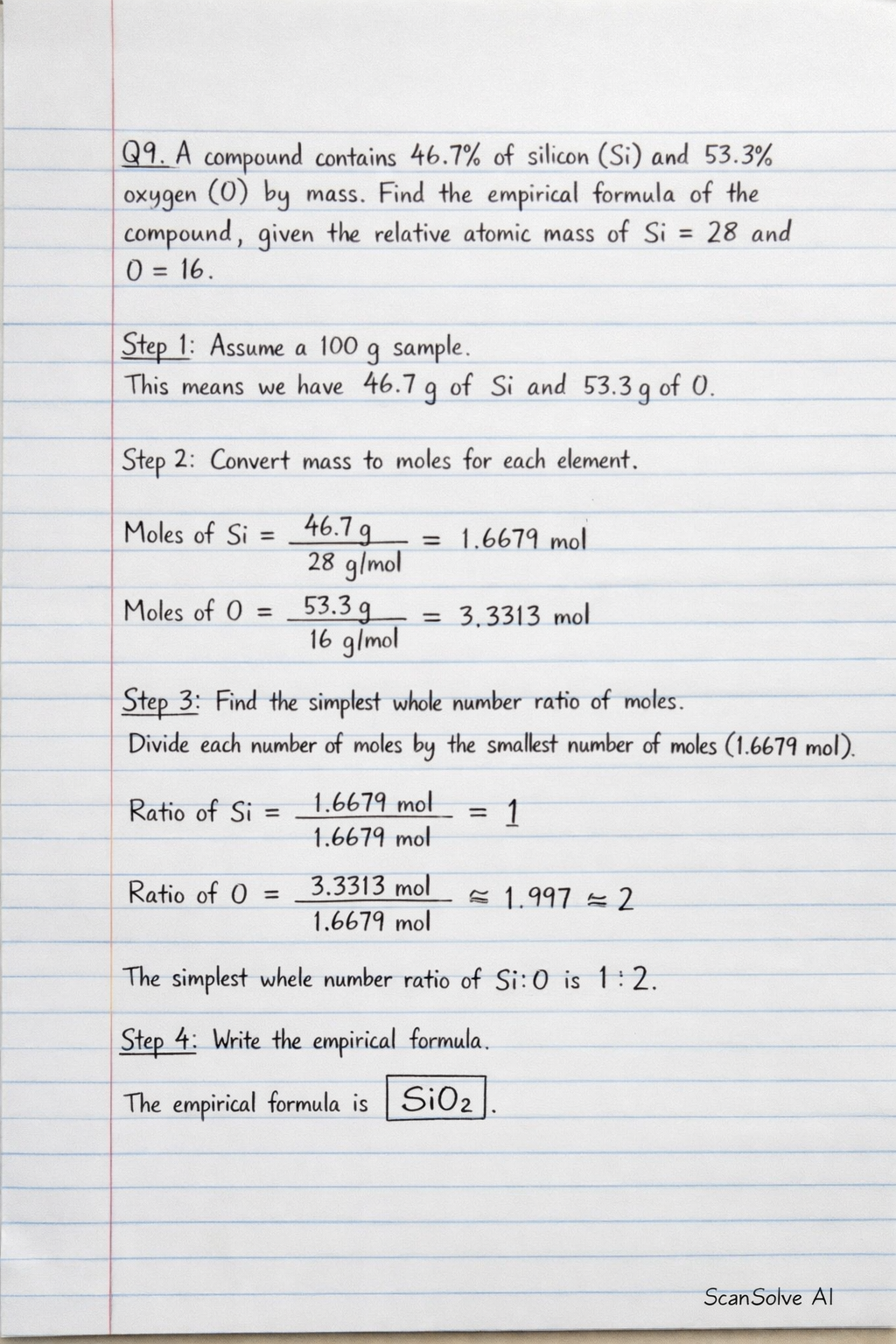
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Q9. A compound contains 46.7% of silicon (Si) and 53.3% oxygen (O) by mass.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.