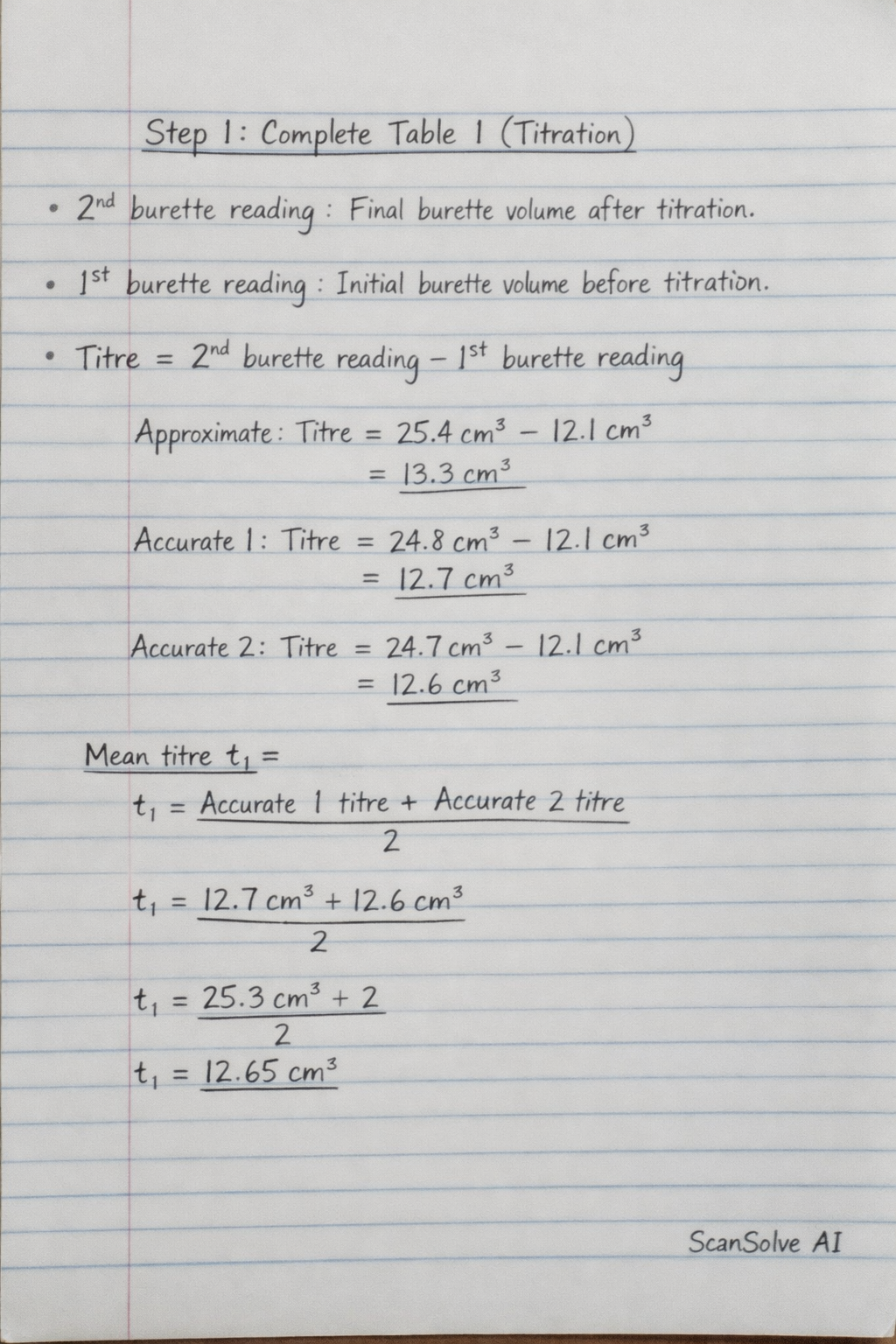To complete the tables, you would follow the experimental procedures and record your measurements. Since no experimental data is provided, I will explain how to fill each table and calculate the required values.
Step 1: Complete Table 1 (Titration)
- 2nd burette reading/ cm³: Record the final volume reading on the burette after the titration reaches the endpoint.
- 1st burette reading/ cm³: Record the initial volume reading on the burette before starting the titration.
- Titre: Calculate the volume of solution R used by subtracting the initial reading from the final reading.
Titre=2ndburettereading−1stburettereading
Perform this calculation for the Approximate, Accurate 1, and Accurate 2 trials.
- Mean titre t1 =: Calculate the average of the accurate titre values (usually Accurate 1 and Accurate 2, if they are concordant, meaning they are very close to each other, e.g., within ±0.1cm3).
Meantitret1=2Accurate1titre+Accurate2titre
The final mean titre should be recorded to two decimal places.
Step 2: Complete Table 2 (Thermochemistry)
- Volume of A in cm³: Record the volume of solution R used for each experiment number.
- Volume of B in cm³: Record the volume of solution S used for each experiment number.
- Temperature of A in °C: Record the initial temperature of solution R for each experiment.
- Temperature of B in °C: Record the initial temperature of solution S for each experiment.
- Highest temperature in °C: Record the maximum temperature reached after mixing solutions R and S for each experiment.
- Temperature change ΔT/°C: Calculate the change in temperature for each experiment. This is the difference between the highest temperature reached and the average of the initial temperatures of solutions A and B.
ΔT=Highesttemperature−(2TemperatureofA+TemperatureofB)
Record ΔT to one decimal place.
Without the actual experimental readings, the tables cannot be filled with numerical values.
Send me the next one 📸