This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
approximately $6
Hey Shãdø, good to see you again.
Here are the definitions for the terms in chemistry:
Avogadro's Constant: Avogadro's constant () is the number of constituent particles (atoms, molecules, ions, or other particles) per mole of a substance. Its value is approximately .
Relative atomic mass: The relative atomic mass () of an element is the ratio of the average mass of atoms of that element to of the mass of an atom of carbon-12. It is a dimensionless quantity.
Relative molecular mass: The relative molecular mass () of a compound is the ratio of the average mass of molecules of that compound to of the mass of an atom of carbon-12. It is also a dimensionless quantity.
The empirical formula: The empirical formula of a chemical compound is the simplest whole-number ratio of atoms of each element present in the compound. It indicates the proportional relationship of elements in a compound.
Send me the next one 📸
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question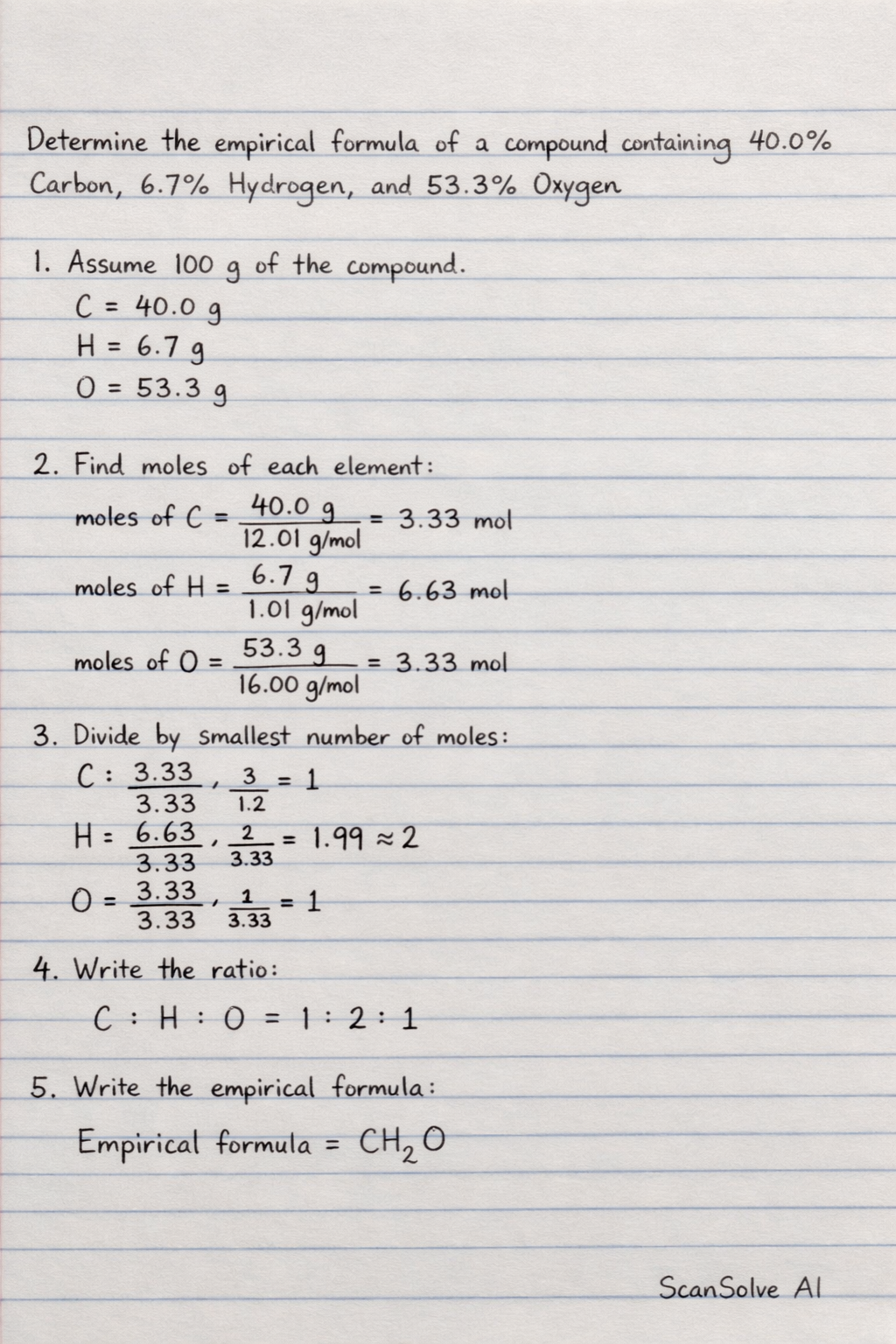
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Hey Shãdø, good to see you again. Here are the definitions for the terms in chemistry: 3) Avogadro's Constant: Avogadro's constant (N_A) is the number of constituent particles (atoms, molecules, ions, or other particles) per mole of a substance.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.