This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
$5
Step 1: Calculate the of benzoic acid. The for benzoic acid () is .
Step 2: Use the Henderson-Hasselbalch equation to find the ratio of the conjugate base to the weak acid. The target is .
Step 3: Calculate the initial moles of sodium benzoate (). The volume of sodium benzoate solution is and its concentration is .
Step 4: Calculate the required moles of benzoic acid (). Since the volume is the same for both components in the buffer, the ratio of concentrations is equal to the ratio of moles.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question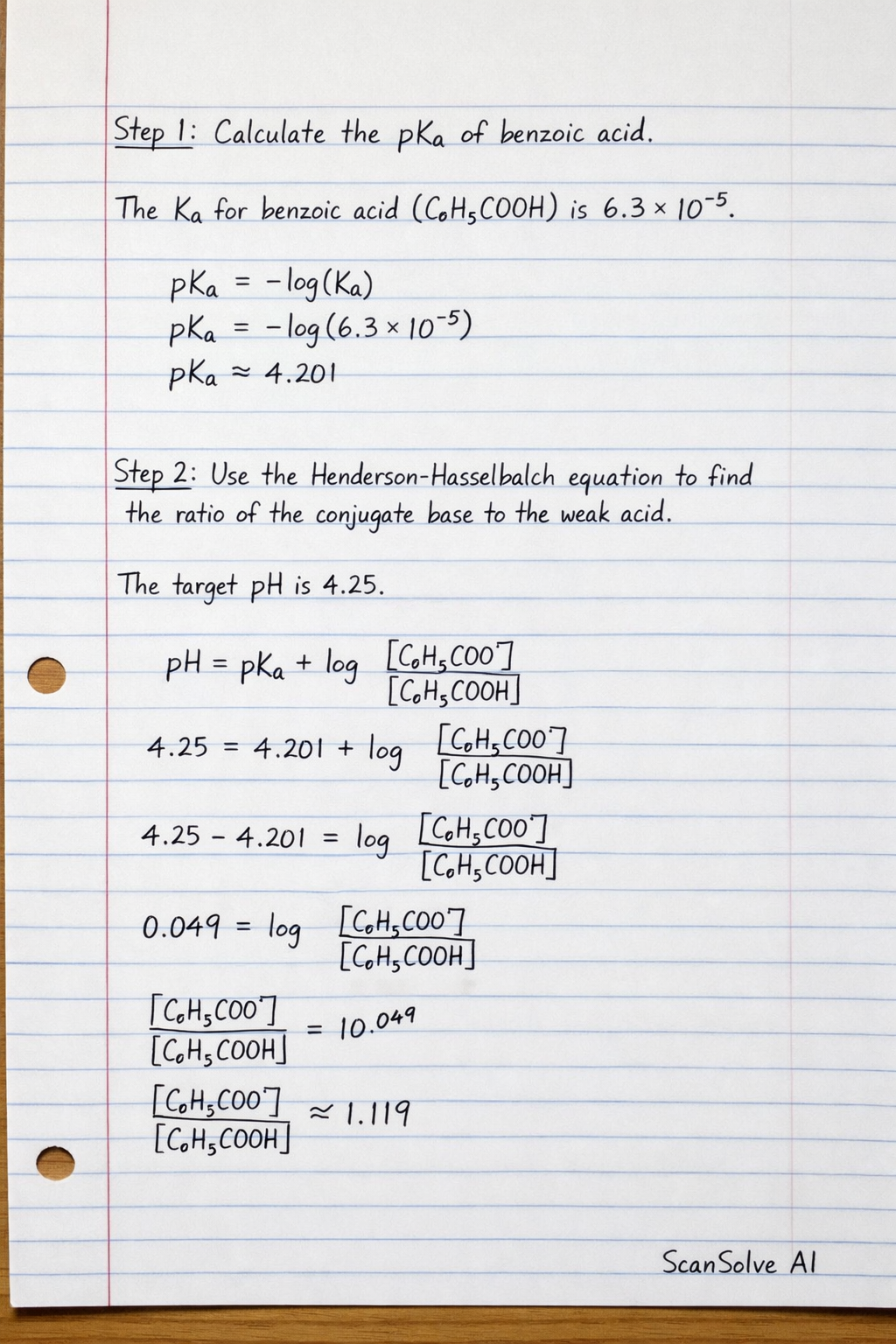
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Calculate the pK_a of benzoic acid. The K_a for benzoic acid (C_6H_5COOH) is 6.3 × 10^-5.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.