This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Here are the missing words for the paragraph:
The periodic table has (i) 18 groups of elements, plus a block of (ii) transition metals. The groups are numbered. Group 4 contains the elements carbon (C), silicon (Si), germanium (Ge), Tin (Sn) and lead (Pb). Their atoms each have 4 electrons in the outer shell. The atoms of group 5 elements each have 5 electrons in the outer shell and so on. Some of the groups have special names too. Group 1 is often called the (iii) alkali metals, group 2 is the alkaline earth metals, group 7 is the (iv) halogens and group 0 is the (v) noble gases.
The horizontal rows in the table are called (vi) periods. Period 2 contains lithium (Li), beryllium (Be), boron (B), carbon (C), nitrogen (N), oxygen (O), fluorine (F) and neon (Ne). There's a zigzag line through the groups. It separates the metals from the non-metals. The (vii) metals are on the left.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question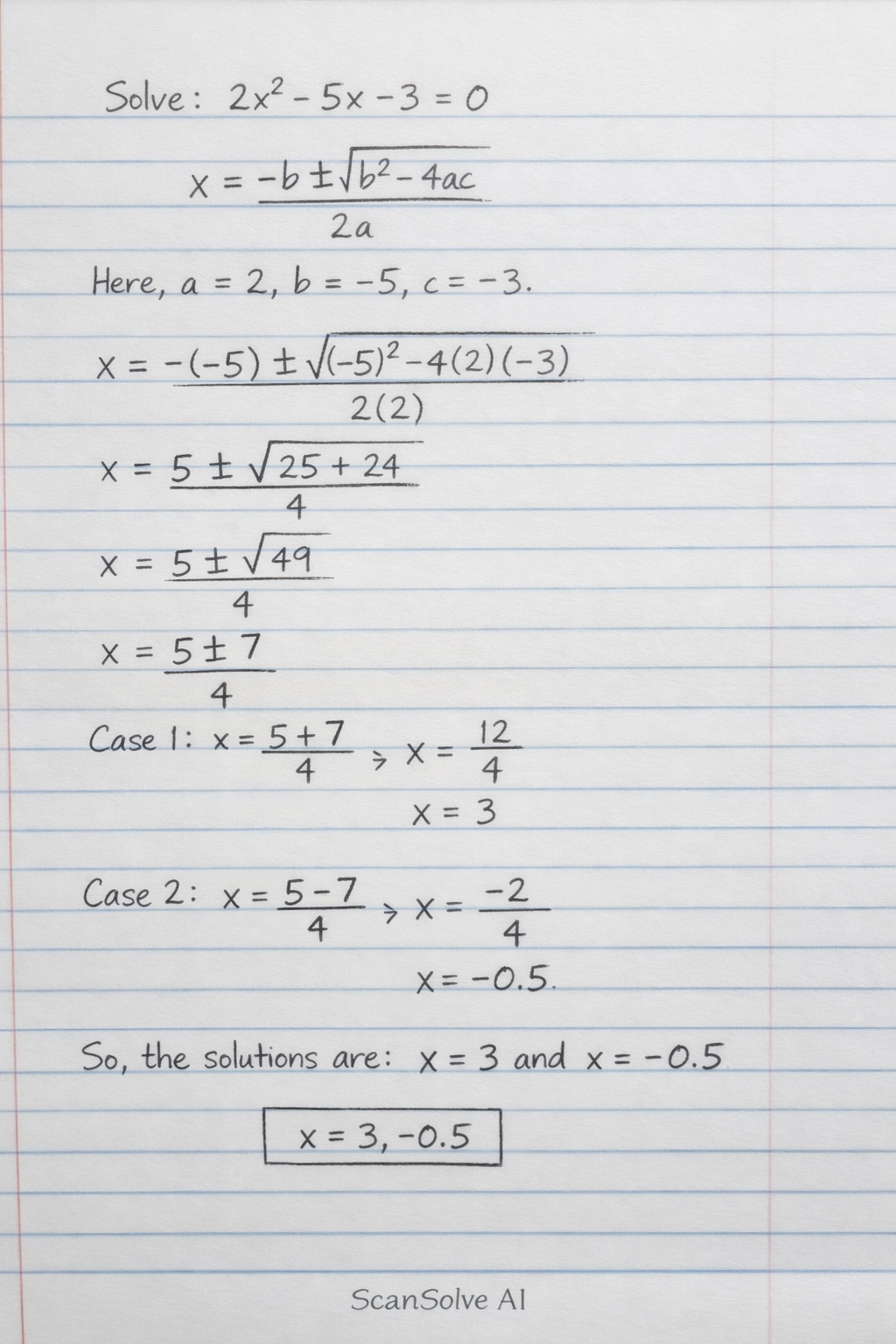
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
The periodic table has (i) 18 groups of elements, plus a block of (ii) transition metals.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.