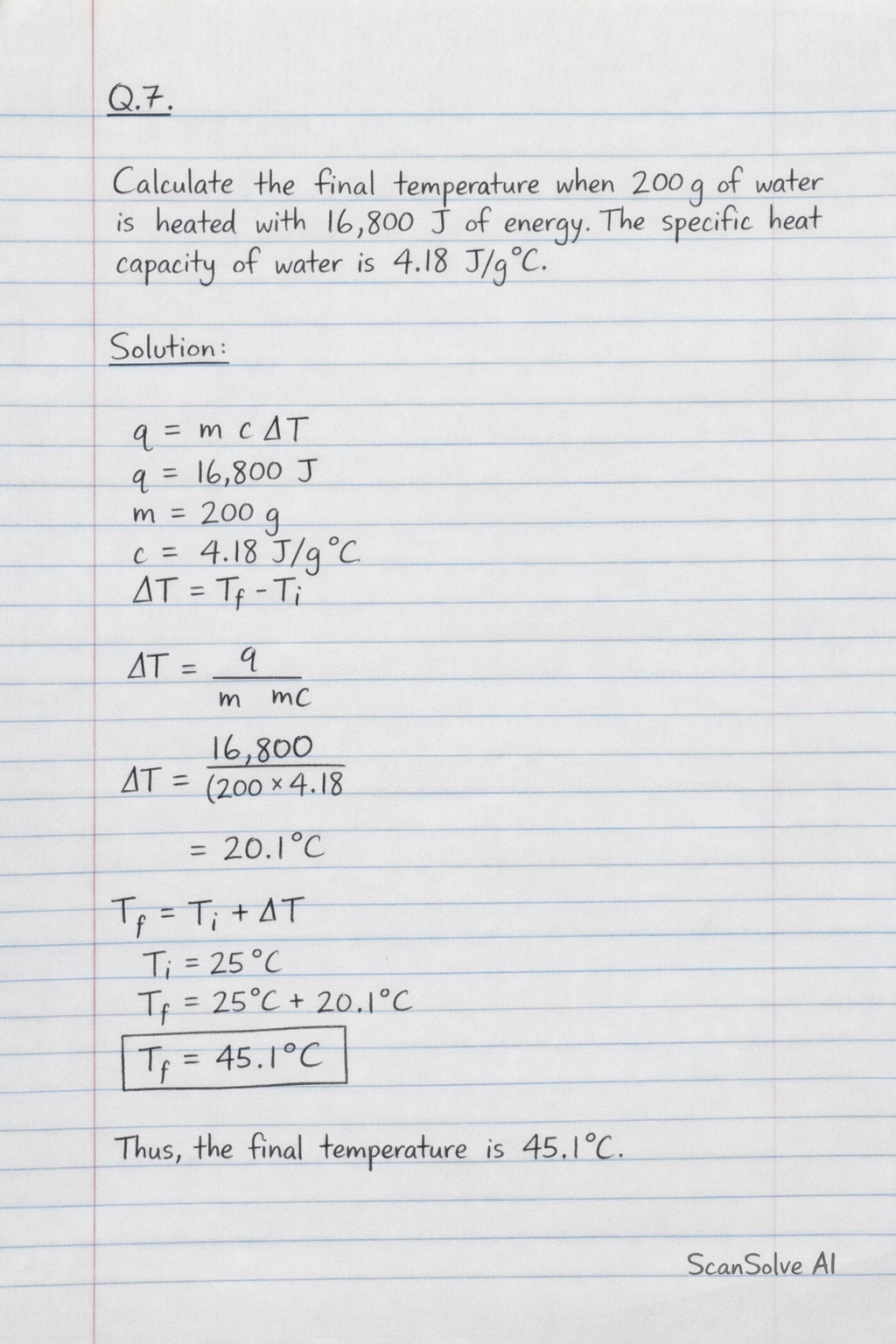This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.
ScanSolve AI Answer
Q7. 1. We need to mention 'atmospheric pressure' in the definition of boiling point because the boiling point is the temperature at which a liquid's vapor pressure equals the external pressure. Atmospheric pressure is the standard external pressure, so it must be specified for a consistent definition. 2. If the pressure is less than atmospheric pressure, water will boil at a lower temperature. This occurs because less vapor pressure is required to overcome the reduced external pressure, allowing boiling to happen at a lower temperature. That's 2 down. 3 left today — send the next one.