This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.
ScanSolve AI Answer
VerifiedStep 1: Convert the time from minutes to seconds.
Step 2: Calculate the total quantity of charge () passed using the formula .
Step 3: Determine the moles of electrons () transferred using Faraday's constant ().
Step 4: Use the stoichiometry of the copper deposition reaction to find the moles of copper formed. The reduction of copper ions is: This means 2 moles of electrons are required to deposit 1 mole of copper. The moles of copper formed is .
That's 2 down. 3 left today — send the next one.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question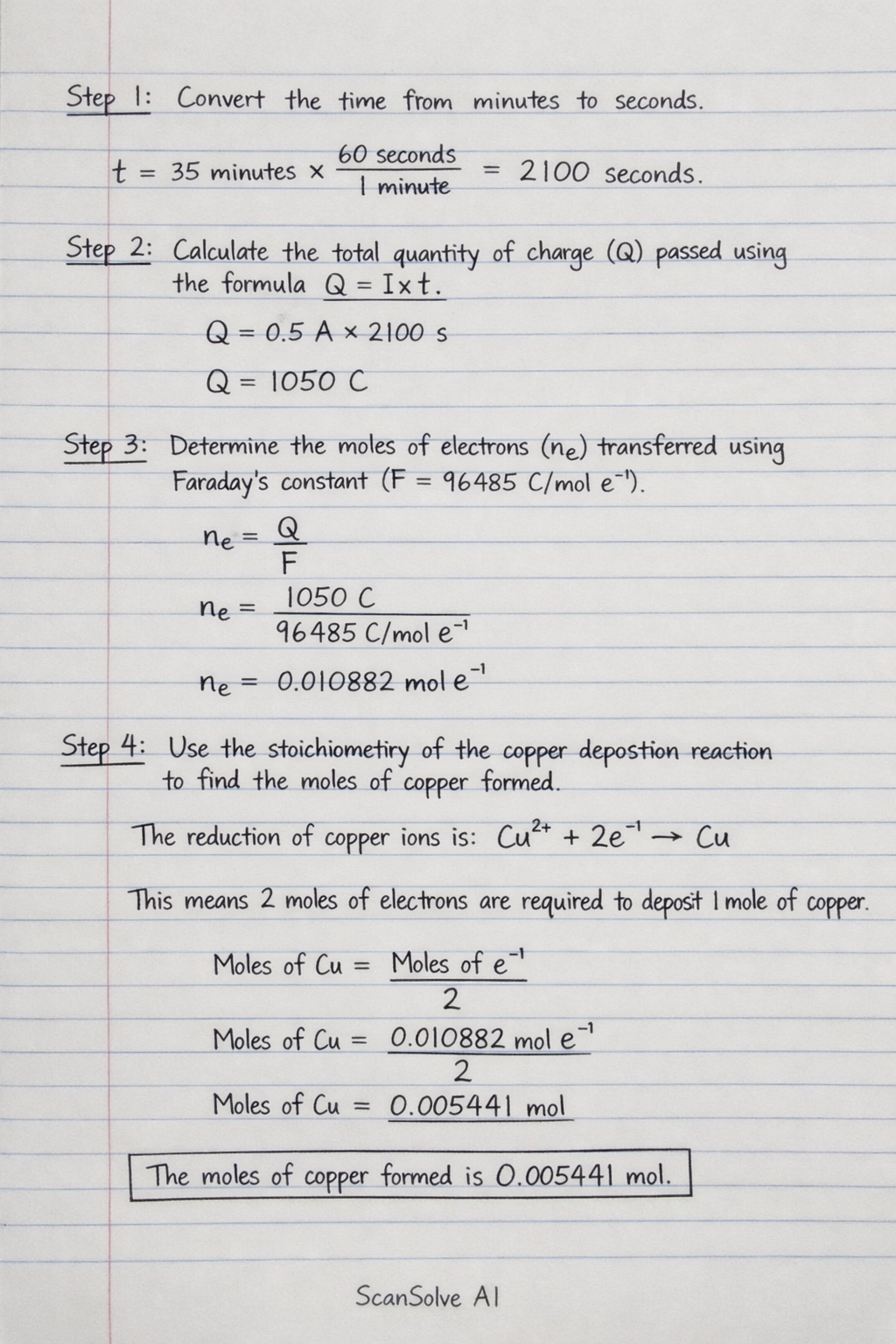
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Convert the time from minutes to seconds. t = 35 minutes × 60 seconds1 minute = 2100 seconds Step 2: Calculate the total quantity of charge (Q) passed using the formula Q = I × t.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.