This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.
ScanSolve AI Answer
Right belackırine@gmail.com, let's go. A dehydration reaction is a type of condensation reaction where two molecules combine to form a larger molecule, with the loss of a molecule of water.
For example, the formation of a disaccharide like maltose from two glucose molecules:
Send me the next one 📸
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question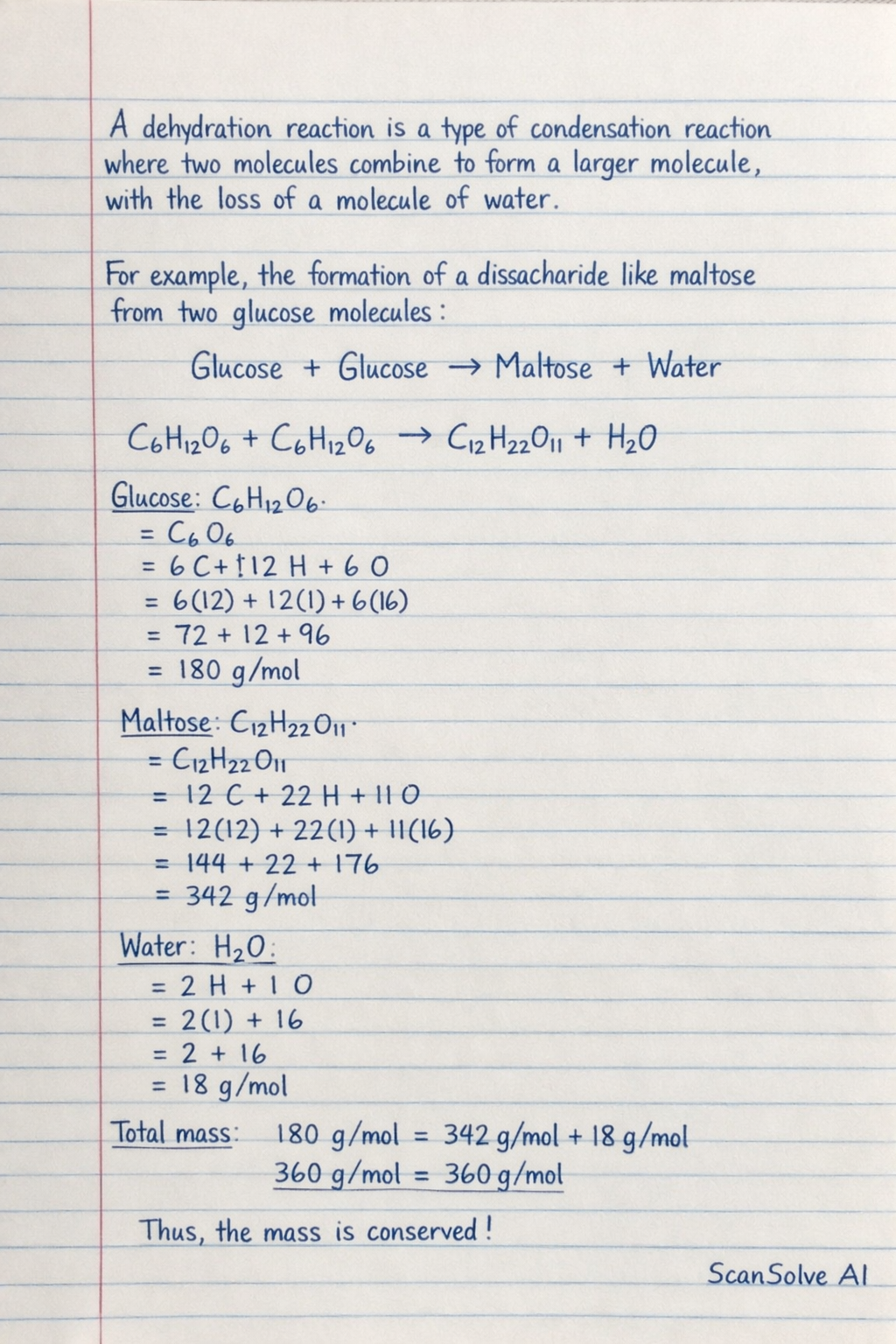
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Right belackırine@gmail.com, let's go. A dehydration reaction is a type of condensation reaction where two molecules combine to form a larger molecule, with the loss of a molecule of water.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.