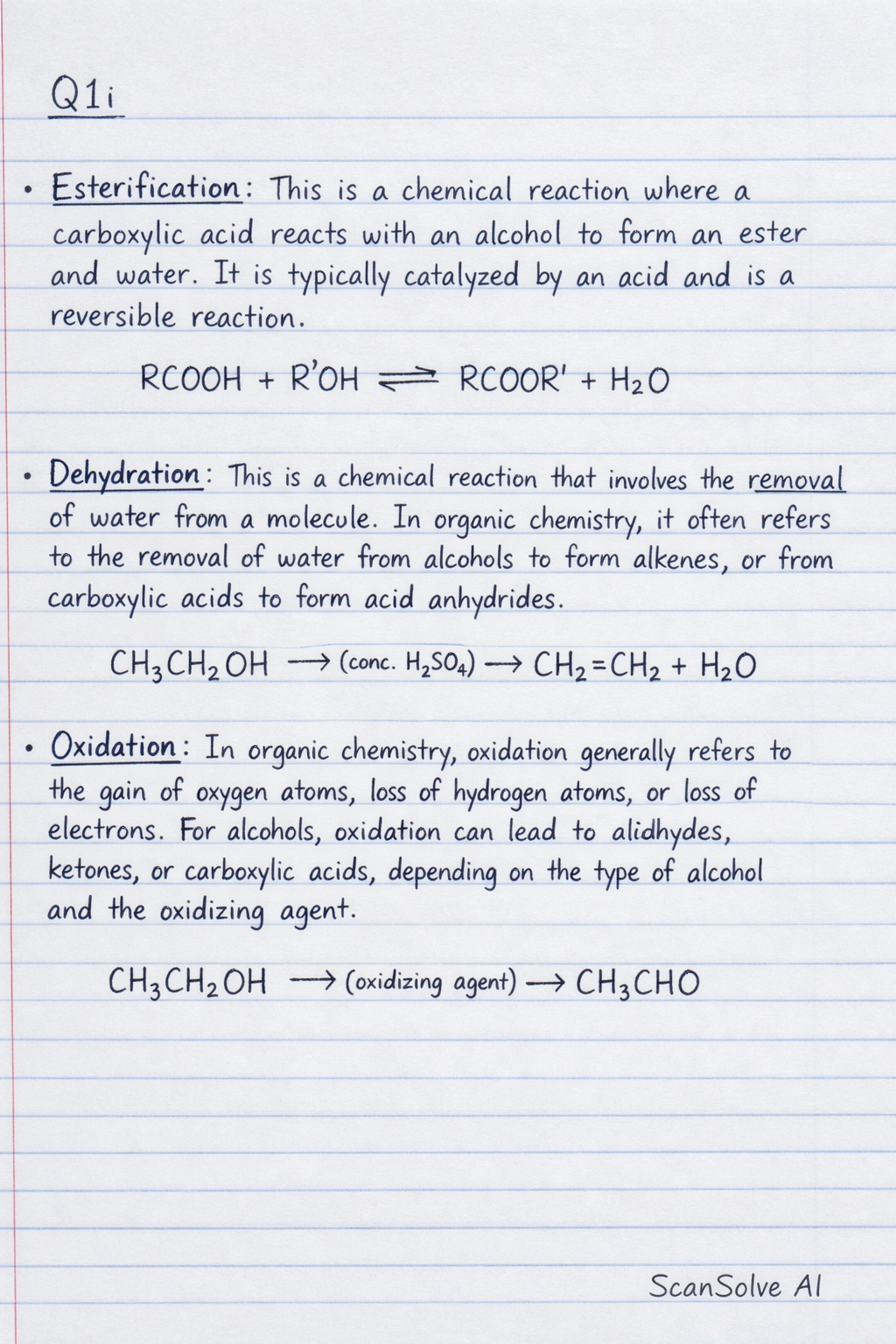This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.
ScanSolve AI Answer
Q1i Esterification: This is a chemical reaction where a carboxylic acid reacts with an alcohol to form an ester and water*. It is typically catalyzed by an acid and is a reversible reaction. RCOOH + R'OH RCOOR' + H_2O Dehydration: This is a chemical reaction that involves the removal of water* from a molecule. In organic chemistry, it often refers to the removal of water from alcohols to form alkenes, or from carboxylic acids to form acid anhydrides. CH_3CH_2OH conc. H_2SO_4 CH_2=CH_2 + H_2O Oxidation: In organic chemistry, oxidation generally refers to the gain of oxygen atoms, loss of hydrogen atoms, or loss of electrons*. For alcohols, oxidation can lead to aldehydes, ketones, or carboxylic acids, depending on the type of alcohol and the oxidizing agent. CH_3CH_2OH oxidizing agent CH_3CHO Q1b Acidity of ROH (Alcohols): Alcohols (ROH) are very weak acids, even weaker than water. They can donate a proton (H^+) to a very strong base, forming an alkoxide ion* (RO^-). The acidity of alcohols decreases as the alkyl group becomes larger due to the electron-donating effect of alkyl groups, which destabilizes the alkoxide ion. ROH + B^- RO^- + BH Metal Alkoxide Formation: Alcohols react with active metals (like sodium or potassium) to form metal alkoxides* and hydrogen gas. This reaction demonstrates the acidic nature of alcohols, as the alcohol donates a proton to the metal. 2ROH + 2Na 2RO^-Na^+ + H_2 Salt Formation with Brønsted Acids: Alkoxide ions (RO^-) are very strong Brønsted bases. They readily react with Brønsted acids (proton donors) to form the corresponding alcohol (a salt in the sense of an ionic compound formed from an acid and a base, though here it's the conjugate acid of the alkoxide). RO^- + HA ROH + A^- Q1ci Simple Polyhydroxylalkanes (Diols and Triols): Diols: Ethane-1,2-diol (Ethylene Glycol): HO & - & CH_2 & - & CH_2 & - & OH Propane-1,2-diol: HO & - & CH_2 & - & CH(OH) & - & CH_3 Propane-1,3-diol: HO & - & CH_2 & - & CH_2 & - & CH_2 & - & OH Triols: Propane-1,2,3-triol (Glycerol/Glycerin): HO & - & CH_2 & - & CH(OH) & - & CH_2 & - & OH Q1cii Source of Propane-1,2,3-triol from Hydrolysis of Triglycerides in Soap Manufacture: Propane-1,2,3-triol (glycerol) is a byproduct of the saponification process, which is the alkaline hydrolysis of triglycerides (fats and oils). Triglycerides are esters formed from glycerol and three fatty acid molecules. During soap manufacture, triglycerides are heated with a strong base (like sodium hydroxide). This breaks the ester bonds, releasing the fatty acid salts (which are soap) and glycerol. Triglyceride + 3NaOH heat Glycerol + 3RCOONa (Soap) Q2a Acidic Nature of Phenol: Phenol (C_6H_5OH) is more acidic than alcohols but less acidic than carboxylic acids. Its acidity arises from the resonance stabilization of its conjugate base, the phenoxide ion* (C_6H_5O^-). The negative charge on the oxygen atom of the phenoxide ion can be delocalized into the benzene ring through resonance. This delocalization spreads the charge over multiple atoms, making the phenoxide ion more stable than an alkoxide ion, thus favoring the dissociation of the proton from phenol. C_6H_5OH C_6H_5O^- + H^+ The resonance structures for phenoxide ion show the negative charge delocalized to the ortho and para positions of the benzene ring. Q2aii Directing Ability of -OH in Electrophilic Substitution Reaction on the Benzene Ring of Phenol: The hydroxyl (-OH) group is a strong activating group and an ortho-para director in electrophilic aromatic substitution reactions. This is because the lone pair of electrons on the oxygen atom can be donated into the benzene ring through resonance, increasing the electron density, particularly at the ortho and para positions. This makes these positions more susceptible to attack by electrophiles. Bromination: Phenol reacts very readily with bromine water (aqueous bromine) at room temperature, without a catalyst, to form 2,4,6-tribromophenol*. The reaction is so vigorous that it does not stop at mono-substitution. C_6H_5OH + 3Br_2(aq) C_6H_2Br_3OH + 3HBr Sulphonation: Phenol reacts with concentrated sulfuric acid* to form phenolsulfonic acids. The product distribution is temperature-dependent: At low temperatures (e.g., 25^), ortho-phenolsulfonic acid* is the major product. At higher temperatures (e.g., 100^), para-phenolsulfonic acid* is the major product (thermodynamic control). Nitration: Phenol reacts with dilute nitric acid at room temperature to yield a mixture of ortho-nitrophenol and para-nitrophenol. Due to the strong activating nature of the -OH group, concentrated nitric acid can lead to the formation of 2,4,6-trinitrophenol* (picric acid), which is explosive. C_6H_5OH + HNO_3(dilute) o-nitrophenol + p-nitrophenol + H_2O Q2b Reactions of the -OH group on Phenol: i. Ether Formation: Phenols can be converted into ethers through the Williamson ether synthesis. This involves first converting phenol into its more nucleophilic phenoxide ion by reacting it with a strong base (like NaOH). The phenoxide ion then reacts with a primary haloalkane* (R'X) via an S_N2 mechanism to form an aryl alkyl ether. C_6H_5OH + NaOH C_6H_5O^-Na^+ + H_2O C_6H_5O^-Na^+ + R'X C_6H_5OR' + NaX ii. Esterification: Phenols react with acyl chlorides or acid anhydrides to form esters* (phenyl esters). Unlike alcohols, phenols do not readily react with carboxylic acids directly under typical acid-catalyzed conditions due to the lower nucleophilicity of the phenolic oxygen. C_6H_5OH + RCOCl C_6H_5OCOR + HCl C_6H_5OH + (RCO)_2O C_6H_5OCOR + RCOOH iii. Alcoholysis in Reaction with Haloalkanes: This term is typically used for reactions where an alcohol acts as a nucleophile. In the context of phenol's -OH group reacting with haloalkanes, it primarily refers to the formation of ethers*, as described in part (i) (Williamson ether synthesis). The phenoxide ion (formed from phenol) acts as a nucleophile to displace the halide from the haloalkane. If "alcoholysis" is interpreted as the reaction of the -OH group of phenol with a haloalkane, it is essentially the Williamson ether synthesis where the phenoxide is the "alcoholate" (alkoxide) equivalent.