This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
\text{31.92 cm
Here is the solution for Question 7, based on the most logical interpretation of the incomplete question:
Question 7:
a. What volume of hydrochloric acid (in ) would be required to react The question is incomplete, as it does not specify what the hydrochloric acid reacts with. Based on the context of previous questions involving and the number "60" visible, we will assume the question asks for the volume of required to react with of the solution from Question 6(a)(ii).
Step 1: Use the titration formula to find the volume of acid (). Step 2: Substitute the assumed values. V_A = \text{31.92 cm^3}
That's 2 down. 3 left today — send the next one.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question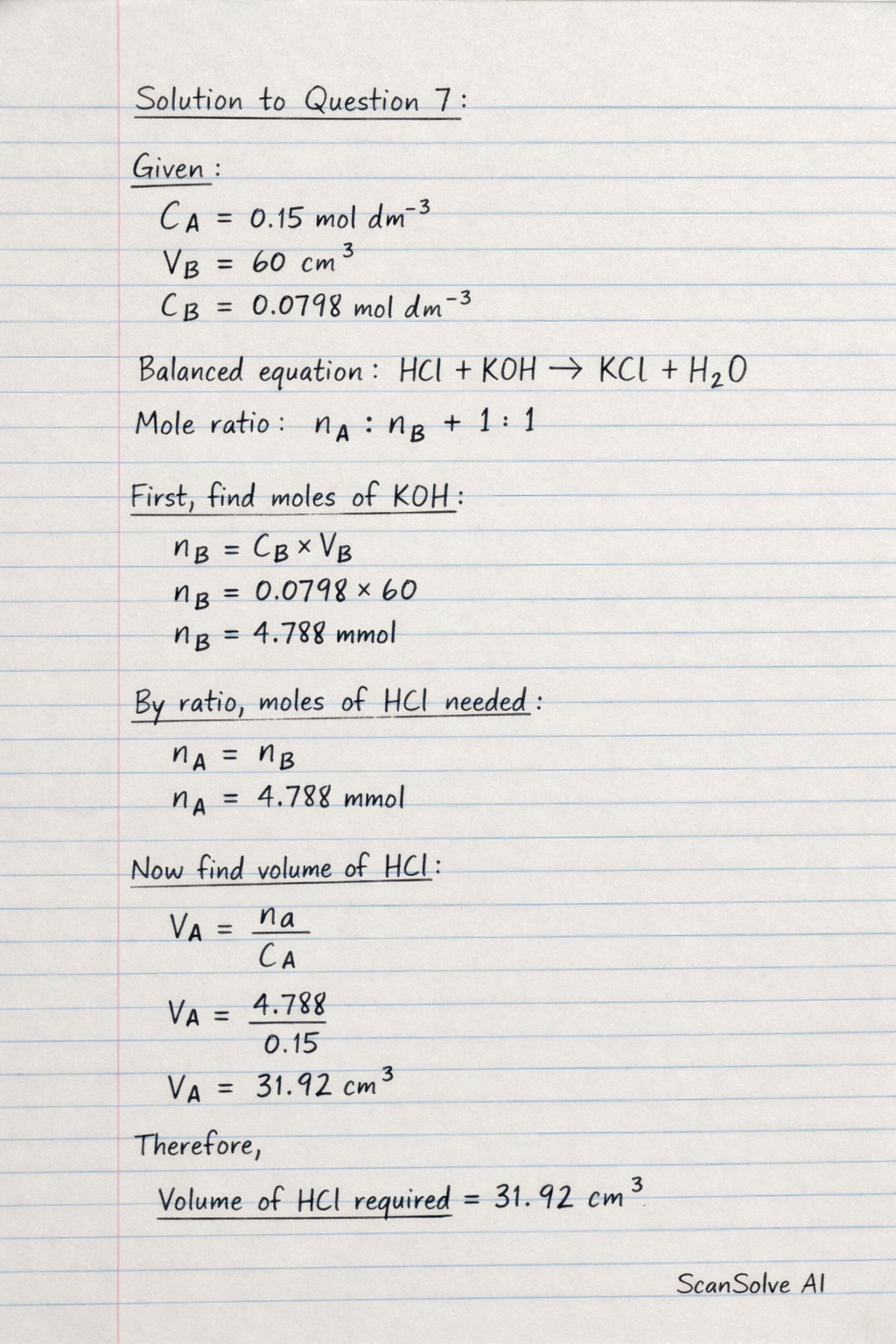
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
Question 7: a. What volume of 0.15 moldm^-3 hydrochloric acid (in cm^3) would be required to react The question is incomplete, as it does not specify what the hydrochloric acid reacts with.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.