This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.

ScanSolve AI Answer
Answer
approximately $1
The formula shown is for the average translational kinetic energy of a molecule in an ideal gas.
Here: • represents the average translational kinetic energy of a single molecule. • is the Boltzmann constant, which relates the average kinetic energy of particles in a gas to the temperature of the gas. Its value is approximately . • is the absolute temperature of the gas in Kelvin.
This formula is correct for the average translational kinetic energy of a molecule in a monatomic ideal gas.
Send me the next one 📸
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question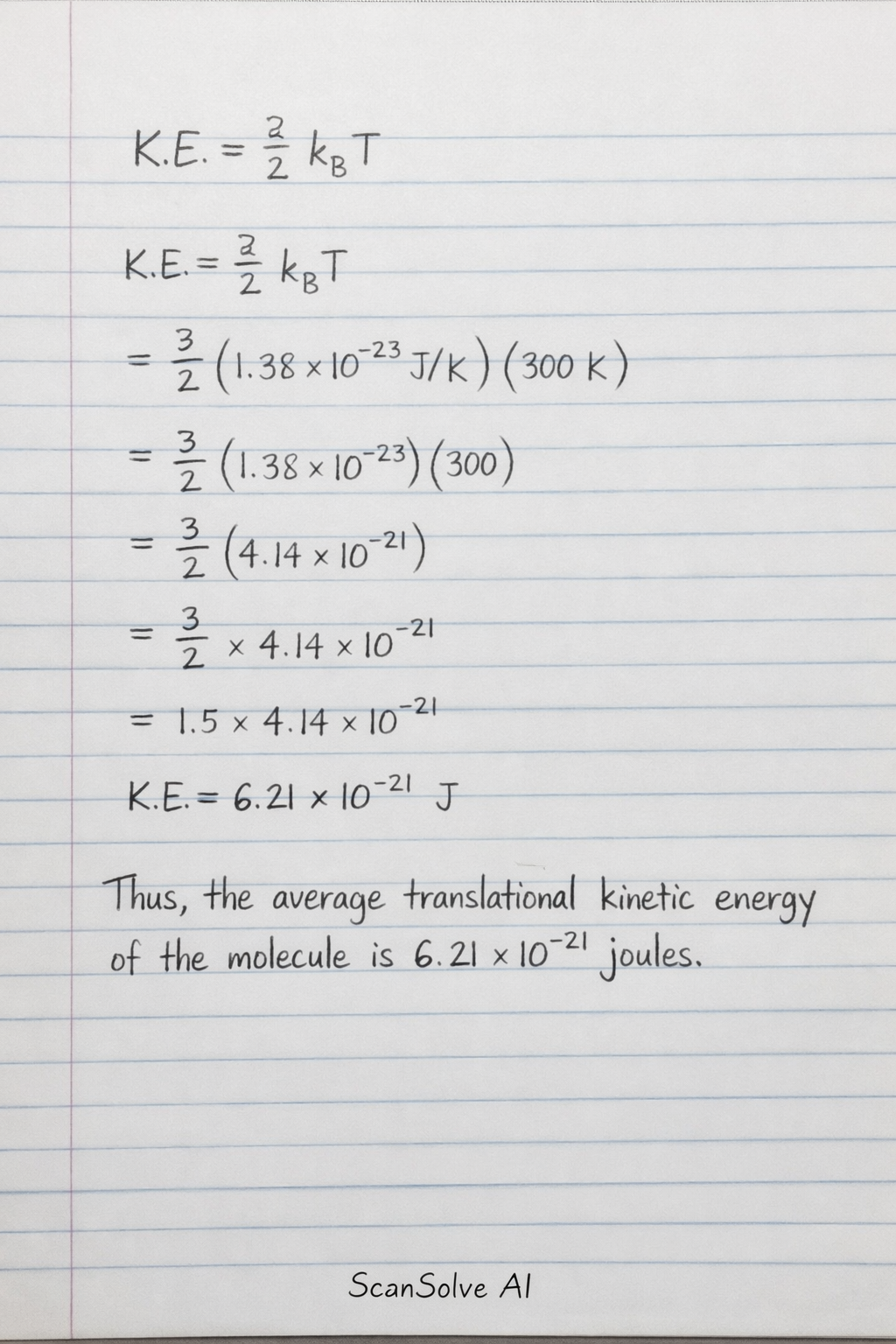
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
The formula shown is for the average translational kinetic energy of a molecule in an ideal gas.
This physics problem tests your understanding of fundamental physical laws and their applications. The step-by-step solution below breaks down the problem using relevant equations and physical reasoning.