This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
Here are the solutions to the chemistry questions:
b) Balance the following chemical equation
i) H + O HO Step 1: Balance oxygen atoms. There are 2 oxygen atoms on the left and 1 on the right. Place a coefficient of 2 in front of HO. Step 2: Balance hydrogen atoms. Now there are 2 hydrogen atoms on the left and on the right. Place a coefficient of 2 in front of H. Step 3: Verify all atoms are balanced. Reactants: H = 4, O = 2 Products: H = 4, O = 2 The equation is balanced. 2\text{H_{2(g)} + O_{2(g)} \rightarrow 2H_{2}O_{(l)}}
ii) KClO KCl + O Step 1: Balance oxygen atoms. There are 3 oxygen atoms on the left and 2 on the right. The least common multiple of 3 and 2 is 6. Place a coefficient of 2 in front of KClO and 3 in front of O. Step 2: Balance potassium and chlorine atoms. Now there are 2 potassium and 2 chlorine atoms on the left, but only 1 of each on the right. Place a coefficient of 2 in front of KCl. Step 3: Verify all atoms are balanced. Reactants: K = 2, Cl = 2, O = 6 Products: K = 2, Cl = 2, O = 6 The equation is balanced. 2\text{KClO_{3(s)} \rightarrow 2KCl_{(s)} + 3O_{2(g)}}
iii) KCO + HCl KCl + CO + HO Step 1: Balance potassium atoms. There are 2 potassium atoms on the left and 1 on the right. Place a coefficient of 2 in front of KCl. Step 2: Balance chlorine atoms. Now there is 1 chlorine atom on the left and 2 on the right. Place a coefficient of 2 in front of HCl. Step 3: Verify all atoms are balanced. Reactants: K = 2, C = 1, O = 3, H = 2, Cl = 2 Products: K = 2, C = 1, O = 2 (from CO) + 1 (from HO) = 3, H = 2, Cl = 2 All atoms are balanced. \text{K_{2}CO_{3(s)} + 2HCl_{(aq)} \rightarrow 2KCl_{(aq)} + CO_{2(g)} + H_{2}O_{(l)}}
10 (a) Construct the Diagram to show the electronic structure in each of the following compounds
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question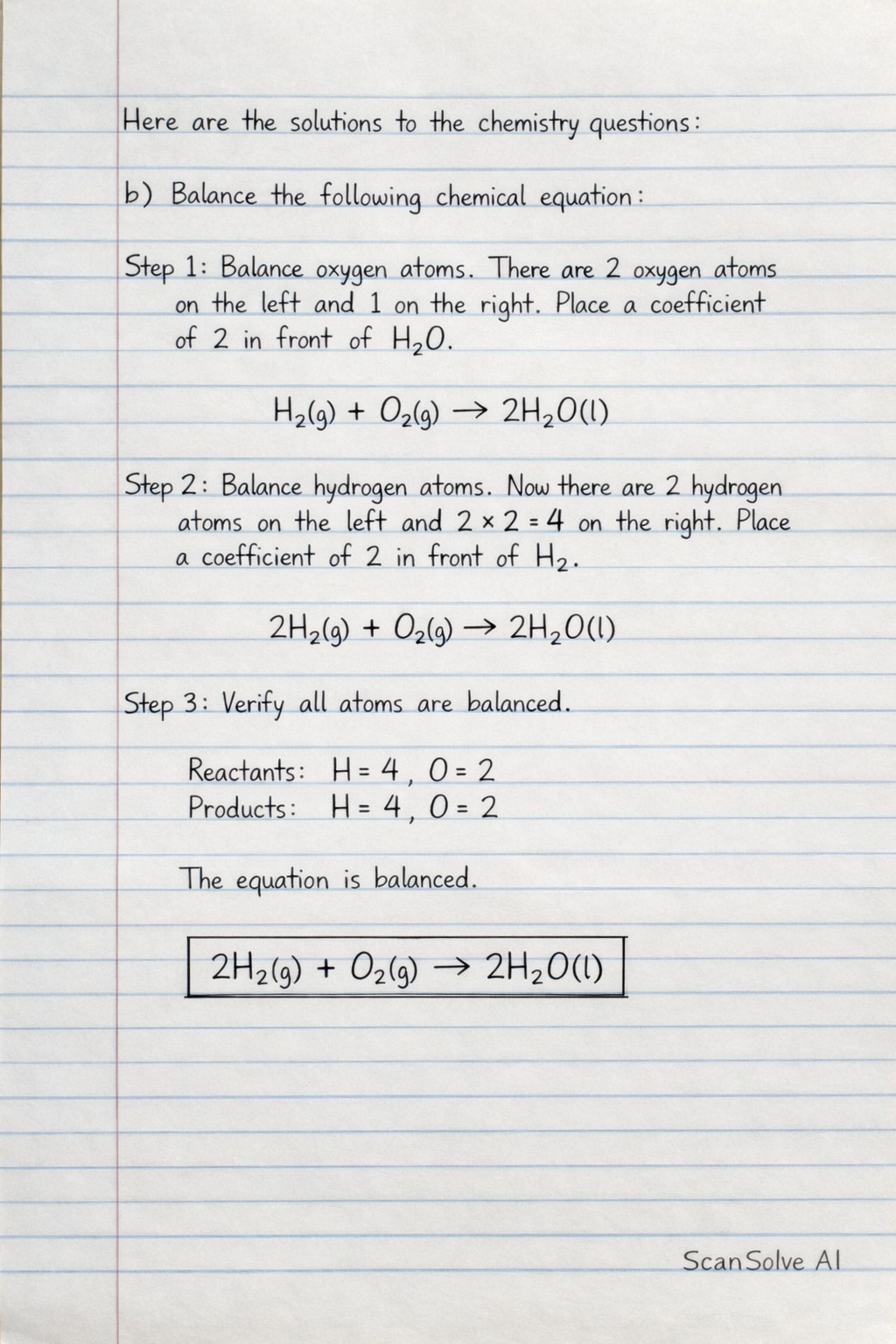
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
b) Balance the following chemical equation i) H_2(g) + O_2(g) → H_2O_(l) Step 1: Balance oxygen atoms.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.