This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
0
Here are the answers to the questions:
b) pH of 13 than pH of 8? pH 13 is more basic (or a stronger base) than pH 8. The student's answer "stronger" is correct in this context.
5) Place the following labels on the pH scale below:
Here is how the labels should be placed on the pH scale:
Far left end of the scale (where the student wrote '7'):
Middle of the scale (at '7'):
Far right end of the scale (at '14'):
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question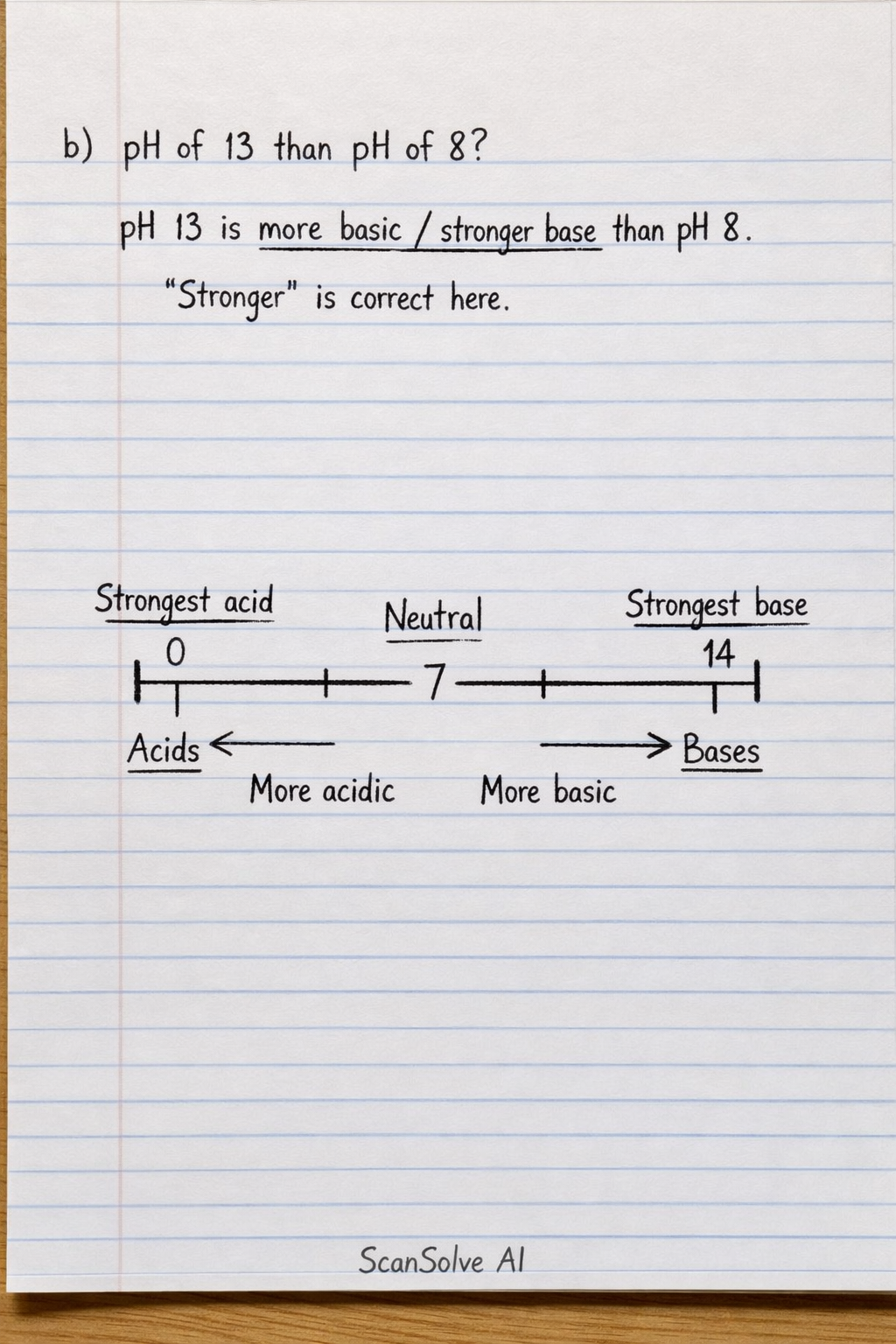
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
b) pH of 13 than pH of 8? pH 13 is more basic (or a stronger base) than pH 8. The student's answer "stronger" is correct in this context.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.