This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.
ScanSolve AI Answer
10 stepsAnswer
3-methylbutan-1-ol
Step 1: Identify the given compound. Let's analyze the structure provided in the image: This can be redrawn to clearly show the carbon chain and substituents: To name this compound:
Step 2: Determine the type of alcohol that oxidizes to an aldehyde. • Primary alcohols () oxidize to aldehydes (). • Secondary alcohols () oxidize to ketones (). • Tertiary alcohols () generally do not oxidize under typical conditions.
Since the product is an aldehyde (3-methylbutanal), it must have been formed from the oxidation of a primary alcohol.
Step 3: Deduce the structure and name of the primary alcohol. To find the corresponding primary alcohol, we replace the aldehyde group () with a primary alcohol group (). So, the structure of the alcohol is: To name this alcohol:
Comparing this with typical multiple-choice options (which were not provided in this turn but are implied by the question format), 3-methylbutan-1-ol is the correct answer.
The alcohol that would produce 3-methylbutanal upon oxidation is .
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question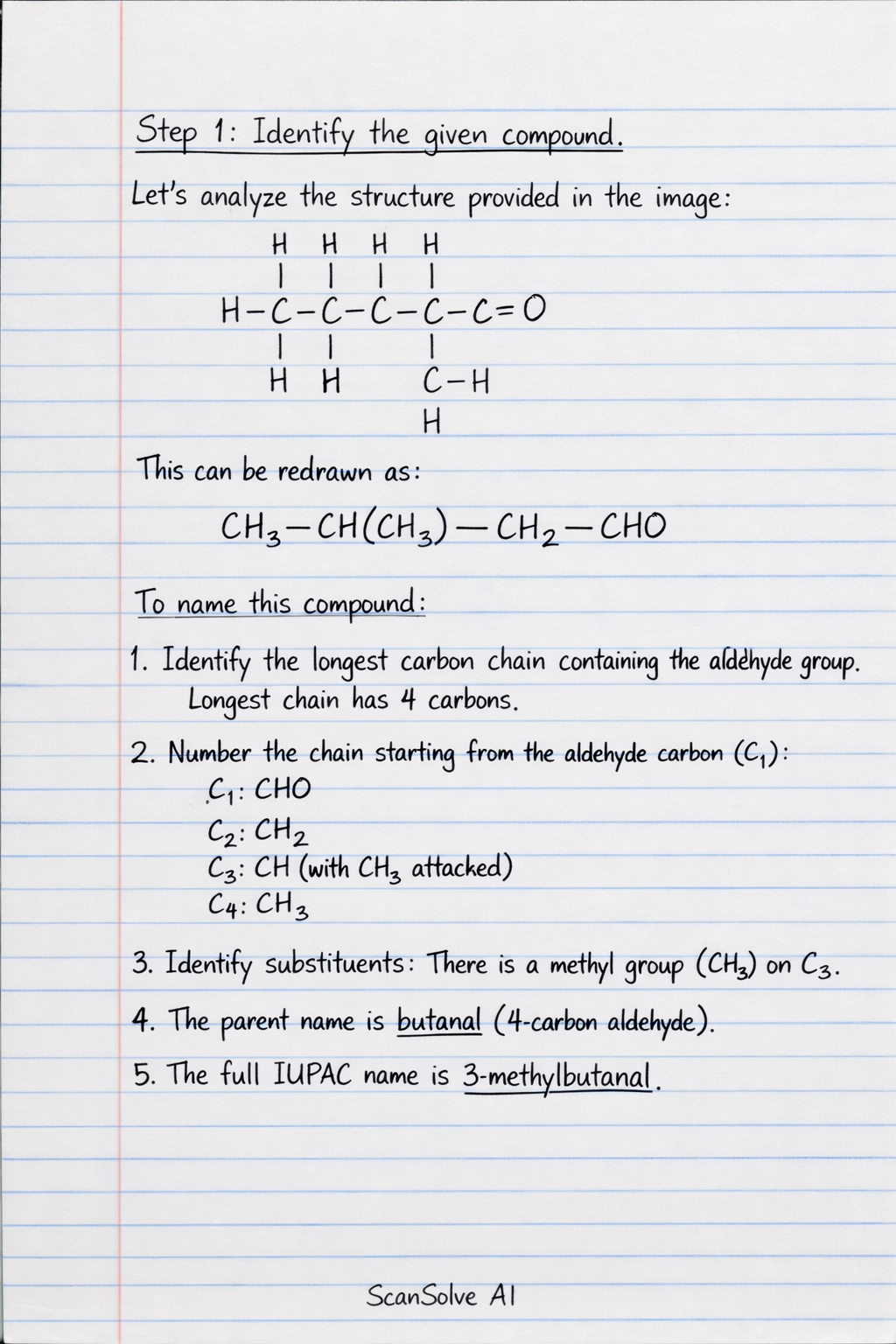
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.