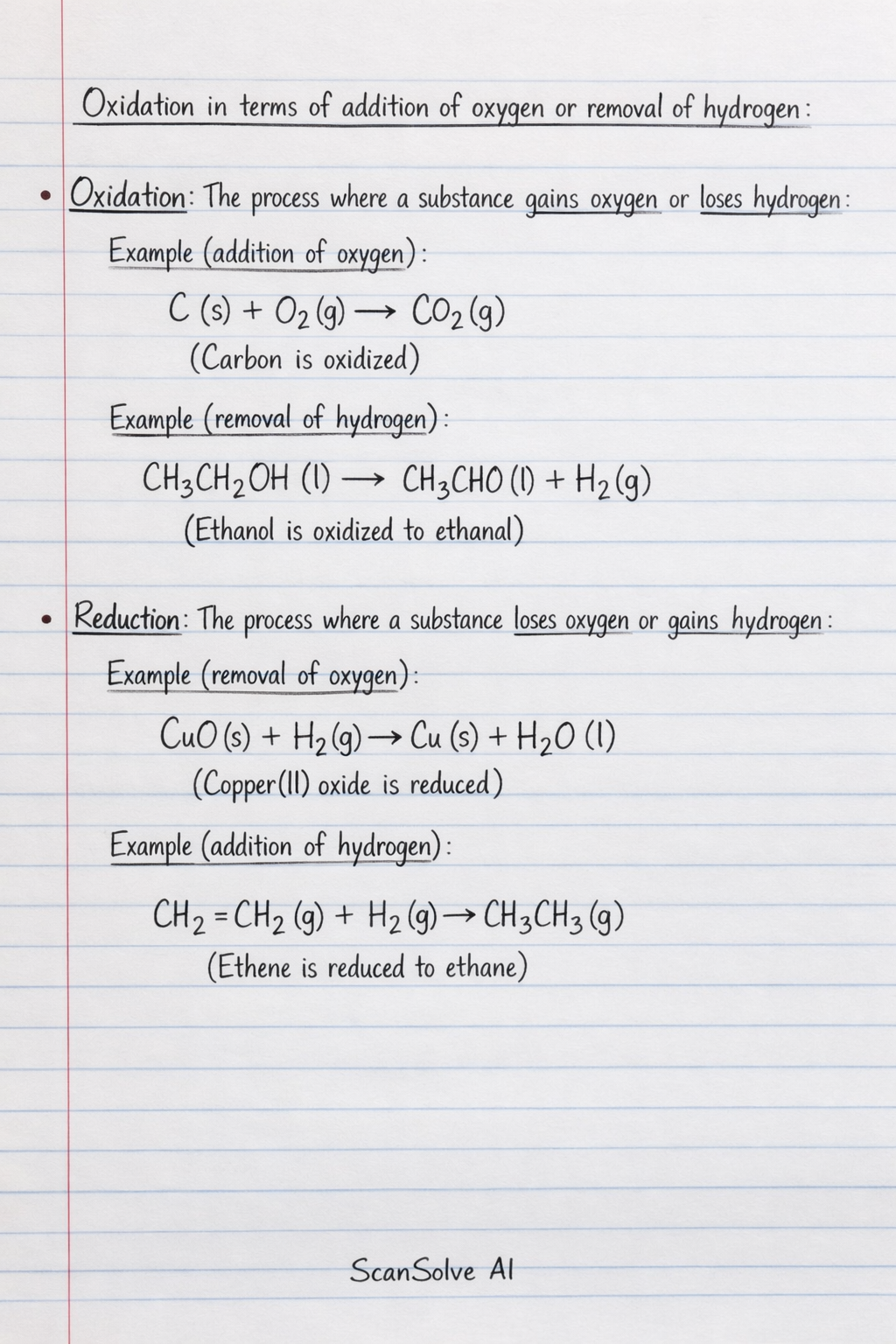Oxidation in terms of addition of oxygen or removal of hydrogen: Oxidation: The process where a substance gains oxygen or loses hydrogen*. Example (addition of oxygen)*: C(s) + O_2(g) CO_2(g) (Carbon is oxidized) Example (removal of hydrogen)*: CH_3CH_2OH(l) CH_3CHO(l) + H_2(g) (Ethanol is oxidized to ethanal) Reduction: The process where a substance loses oxygen or gains hydrogen*. Example (removal of oxygen)*: CuO(s) + H_2(g) Cu(s) + H_2O(l) (Copper(II) oxide is reduced) Example (addition of hydrogen)*: CH_2=CH_2(g) + H_2(g) CH_3CH_3(g) (Ethene is reduced to ethane) Oxidation and reduction in terms of electron transfer: Oxidation: The process of losing electrons*. Reduction: The process of gaining electrons*. Mnemonic*: OIL RIG (Oxidation Is Loss, Reduction Is Gain of electrons). Use of Oxidation Numbers: An oxidation number* (or oxidation state) is a hypothetical charge an atom would have if all bonds were purely ionic. It helps track electron transfer in redox reactions. Rules for assigning oxidation numbers: 1. An atom in its elemental form has an oxidation number of 0 (e.g., O_2, Na, Cl_2). 2. For a monatomic ion, the oxidation number is equal to its charge (e.g., Na^+ is +1, O^2- is -2). 3. Oxygen usually has an oxidation number of -2 in compounds, except in peroxides (e.g., H_2O_2) where it is -1, and with fluorine (e.g., OF_2) where it is +2. 4. Hydrogen usually has an oxidation number of +1 in compounds, except in metal hydrides (e.g., NaH) where it is -1. 5. Group 1 metals are always +1, Group 2 metals are always +2. 6. Fluorine is always -1. Other halogens are usually -1, except when bonded to oxygen or a more electronegative halogen. 7. The sum of oxidation numbers in a neutral compound is 0. 8. The sum of oxidation numbers in a polyatomic ion equals the charge of the ion. Oxidation and reduction treated as change in oxidation numbers: Oxidation: An increase* in oxidation number. Reduction: A decrease* in oxidation number. Use of oxidation numbers in balancing simple equations: Step 1: Assign oxidation numbers to all atoms in the unbalanced equation. Step 2: Identify atoms that undergo a change in oxidation number. Step 3: Determine the total change in oxidation number for the oxidized and reduced species. Step 4: Balance the changes in oxidation numbers by using coefficients in front of the species. Step 5: Balance the remaining atoms (usually oxygen and hydrogen) by inspection, adding H_2O and H^+ (for acidic solutions) or OH^- (for basic solutions). Example (Acidic solution): Balance MnO_4^- + Fe^2+ Mn^2+ + Fe^3+ 1. Assign oxidation numbers: Mn in MnO_4^- is +7. O is -2. Fe^2+ is +2. Mn^2+ is +2. Fe^3+ is +3. 2. Changes: Mn: +7 +2 (decrease of 5, reduction) Fe: +2 +3 (increase of 1, oxidation) 3. Balance changes: To balance the 5 decrease in Mn with the 1 increase in Fe, we need 5 Fe^2+ ions. MnO_4^- + 5Fe^2+ Mn^2+ + 5Fe^3+ 4. Balance oxygen (with H_2O) and hydrogen (with H^+): There are 4 oxygen atoms on the left in MnO_4^-. Add 4H_2O to the right. MnO_4^- + 5Fe^2+ Mn^2+ + 5Fe^3+ + 4H_2O Now there are 8 hydrogen atoms on the right in 4H_2O. Add 8H^+ to the left. 8H^+ + MnO_4^- + 5Fe^2+ Mn^2+ + 5Fe^3+ + 4H_2O (Check charges: Left: 8(+1) + (-1) + 5(+2) = +8 - 1 + 10 = +17. Right: (+2) + 5(+3) + 0 = +2 + 15 = +17. Charges are balanced.) IUPAC nomenclature of inorganic compounds using oxidation number: For compounds containing metals that can have multiple oxidation states (e.g., transition metals), the oxidation number is indicated by a Roman numeral in parentheses immediately following the metal's name. Example*: FeCl_2: Iron(II) chloride (Iron has an oxidation state of +2) FeCl_3: Iron(III) chloride (Iron has an oxidation state of +3) CuO: Copper(II) oxide (Copper has an oxidation state of +2) Cu_2O: Copper(I) oxide (Copper has an oxidation state of +1) Tests for oxidizing and reducing agents: An oxidizing agent* (or oxidant) causes another substance to be oxidized while itself being reduced. A reducing agent* (or reductant) causes another substance to be reduced while itself being oxidized. Tests for Oxidizing Agents: Oxidizing agents typically oxidize* indicator substances, causing a visible color change. Potassium iodide (KI): Oxidizing agents will oxidize I^- ions to I_2 (iodine), which is brown in solution or turns starch indicator blue-black. Example*: MnO_4^- (purple) + I^- (colorless) Mn^2+ (colorless) + I_2 (brown) Acidified potassium manganate(VII) (KMnO_4): Strong oxidizing agents will remain purple or change color less readily than when reacting with a reducing agent. (This is more commonly used to test for reducing agents, as it is itself a strong oxidizing agent). Acidified potassium dichromate(VI) (K_2Cr_2O_7): Strong oxidizing agents will remain orange or change color less readily. Tests for Reducing Agents: Reducing agents typically reduce* indicator substances, causing a visible color change. Acidified potassium manganate(VII) (KMnO_4): Reducing agents will reduce the purple MnO_4^- ion to the colorless Mn^2+ ion. The purple solution will decolorize. Example*: Fe^2+ (pale green) + MnO_4^- (purple) Fe^3+ (yellow-brown) + Mn^2+ (colorless) Acidified potassium dichromate(VI) (K_2Cr_2O_7): Reducing agents will reduce the orange Cr_2O_7^2- ion to the green Cr^3+ ion. The orange solution will turn green. Example*: SO_2 (gas) + Cr_2O_7^2- (orange) SO_4^2- + Cr^3+ (green) Fehling's solution or Benedict's solution: Used to test for reducing sugars (which are reducing agents). A blue solution turns brick-red upon heating in the presence of a reducing agent.