This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
mixture D contains
1 (b) M is an element that belongs to the 3rd period of the periodic table and is a member of the alkaline earth metals. Show the position of M in the grid. The element M is Magnesium (Mg). It is located in Group 2, Period 3 of the periodic table.
2 (a) The diagram below shows spots of pure substances A, B and C on a chromatography paper. Spot D is that of a mixture. After development A, B and C were found to have moved 8cm, 3cm and 6cm respectively. D was separated into two spots which had moved 6cm and 8cm.
(i) On the diagram: (I) Label the baseline (origin) The baseline (origin) is the horizontal line at the bottom of the chromatography paper where the initial spots A, B, C, and D were applied. It is marked by the 'X' symbols.
(II) Show the positions of all the spots after development • Spot A: Mark a spot at the 8 cm line above the origin, directly above the initial 'X' for A. • Spot B: Mark a spot at the 3 cm line above the origin, directly above the initial 'X' for B. • Spot C: Mark a spot at the 6 cm line above the origin, directly above the initial 'X' for C. • Spot D: Mark two spots above the initial 'X' for D: one at the 6 cm line and another at the 8 cm line.
(ii) Identify the substances present in mixture D Mixture D separated into two spots that moved 6 cm and 8 cm. Substance C moved 6 cm. Substance A moved 8 cm. Therefore, mixture D contains substances A and C.
2 (b) Describe how solid ammonium chloride can be separated from a solid mixture of ammonium chloride and anhydrous calcium chloride. This separation can be achieved by sublimation. Step 1: Place the solid mixture of ammonium chloride and anhydrous calcium chloride in an evaporating dish. Step 2: Gently heat the evaporating dish. Ammonium chloride will sublime, turning directly from a solid into a gas. Step 3: Place an inverted funnel or watch glass over the evaporating dish. The ammonium chloride vapor will cool and solidify (deposit) on the cooler surface of the funnel/watch glass, forming pure solid ammonium chloride. Step 4: The anhydrous calcium chloride, which does not sublime, will remain in the evaporating dish.
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question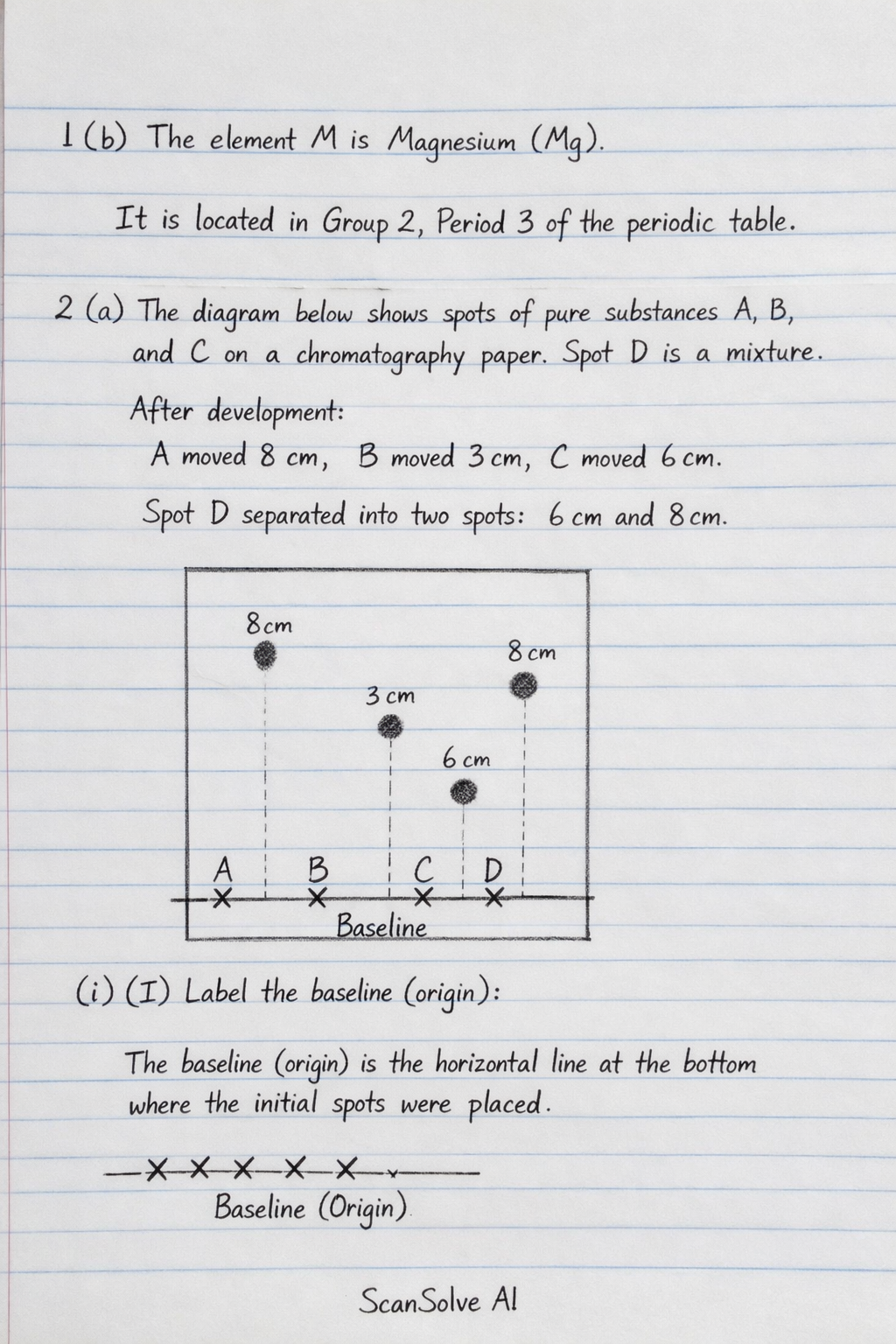
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
1 (b) M is an element that belongs to the 3rd period of the periodic table and is a member of the alkaline earth metals.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.