This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.

ScanSolve AI Answer
Answer
Here are the solutions to the questions:
1. (d) (i) Determine the overall order of the reaction showing all the steps. The general rate law for this reaction can be written as: where is the order with respect to , is the order with respect to , and is the rate constant.
Step 1: Determine the order with respect to (). Compare Experiment 1 and Experiment 2, where is kept constant. From Experiment 1: From Experiment 2: Divide by : Therefore, . The reaction is first order with respect to .
Step 2: Determine the order with respect to (). Compare Experiment 2 and Experiment 3, where is kept constant. From Experiment 2: From Experiment 3: Divide by :
Get instant step-by-step solutions to any question. Free to start.
Ask Your Question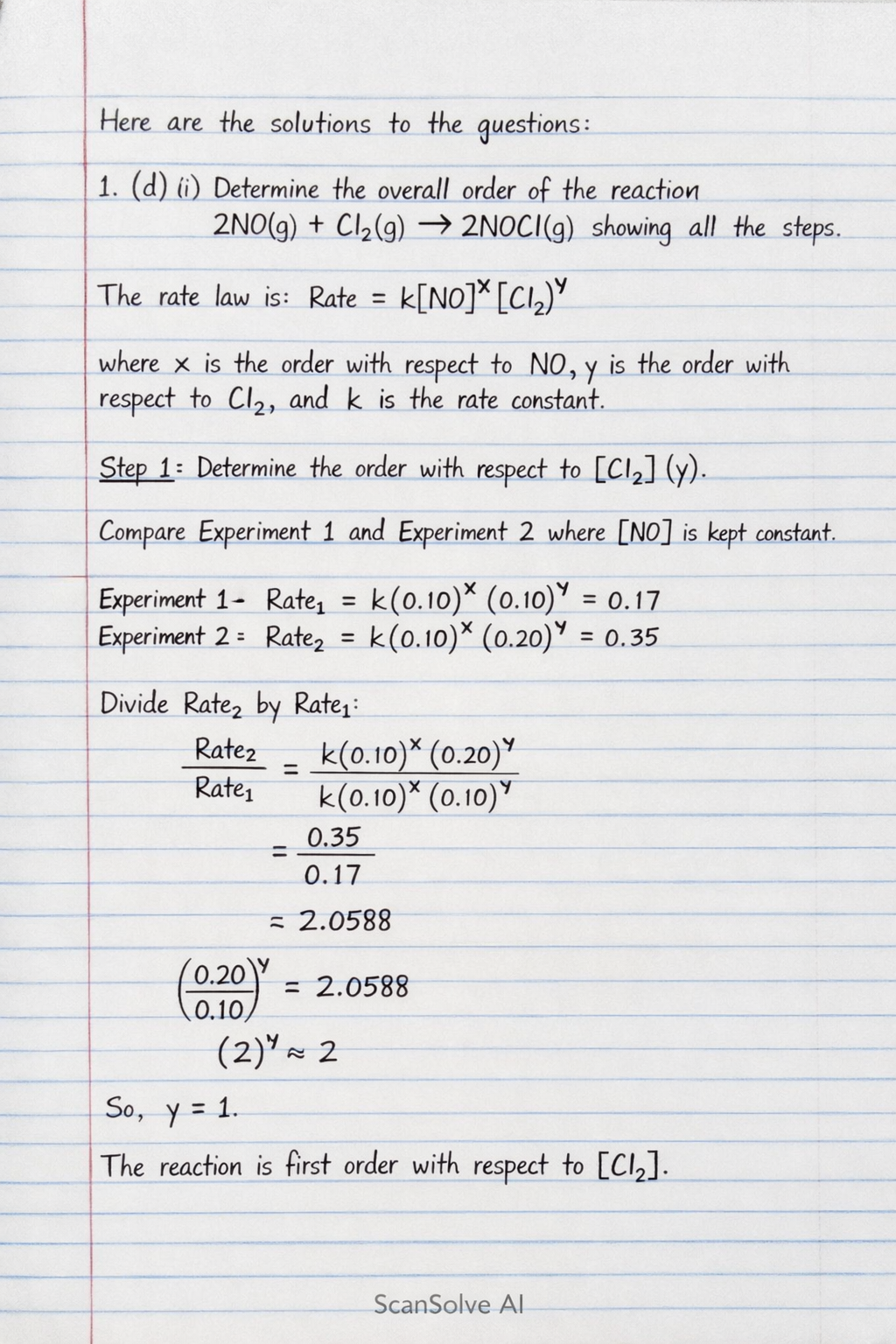
Handwritten Step-by-Step Solution
Get this as a handwritten image on WhatsApp — like a tutor wrote it for you.
Try on WhatsAppStill have questions?
1. (d) (i) Determine the overall order of the reaction 2NO(g) + Cl_2(g) 2NOCl(g) showing all the steps.
This chemistry question involves key chemical concepts and calculations. The detailed solution below walks through each step, from identifying the reaction type to computing the final answer.